Abstract
Rationale
Nonmedical use and abuse of prescription opioids is an increasing public health problem. Intravenous (IV) administration of opioid analgesics intended for oral use is not uncommon; yet, little is known about the relative abuse potential of these drugs when administered intravenously to recreational opioid abusers without physical dependence.
Methods
This inpatient study employed a double-blind, randomized, within-subject, placebo-controlled design to examine the relative abuse potential of IV doses of oxycodone, hydrocodone, and morphine. Nine healthy adult participants reporting recreational opioid use and histories of IV opioid use completed 11 experimental sessions, including one active-dose practice session. IV doses were infused over 5 min and included three identical doses of each opioid (5, 10, and 20 mg/10 ml) and saline placebo. Physiological, subjective, and performance effects were collected before and for 6 h after drug administration.
Results
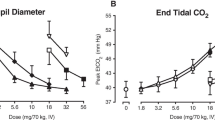
All three opioids produced prototypical mu agonist effects (e.g., miosis; increased ratings of liking) that were generally dose-related. Pharmacodynamic effects were observed within 5 min of IV administration. Physiological effects were more prolonged than subjective effects for all three drugs. While the magnitude of effects was generally comparable across drugs and qualitatively similar, valid potency assays indicated the following potency relationship: oxycodone > morphine > hydrocodone.
Conclusions
There were modest potency differences between oxycodone, hydrocodone, and morphine, but their overall profile of effects was similar, indicating significant abuse potential when administered intravenously.



Similar content being viewed by others
References
Abreu ME, Bigelow GE, Fleisher L, Walsh SL (2001) Effect of intravenous injection speed on responses to cocaine and hydromorphone in humans. Psychopharmacology 154:76–84
Comer SD, Sullivan MA, Whittington RA, Vosburg SK, Kowalczyk WJ (2008) Abuse liability of prescription opioids compared to heroin in morphine-maintained heroin abusers. Neuropsychopharmacology 33:1179–1191
Comer SD, Ashworth JB, Sullivan MA, Vosburg SK, Saccone PA, Foltin RW (2009) Relationship between rate of infusion and reinforcing strength of oxycodone in humans. J Opioid Manag 5:203–212
Davis WR, Johnson BD (2008) Prescription opioid use, misuse, and diversion among street drug users in New York City. Drug Alcohol Depend 92:267–276
Finney DJ (1964) Statistical method in biological assay, 2nd edn. Hafner, New York
Fraser HF, Isbell H (1950) Addiction liabilities of morphinan, 6-methyldihydromorphine and dihydrocodeinone. J Pharmacol Exp Ther 100:128–135
Havens JR, Walker R, Leukefeld CG (2007) Prevalence of opioid analgesic injection among rural nonmedical opioid analgesic users. Drug Alcohol Depend 87:98–102
Hays L, Kirsh KL, Passik SD (2003) Seeking drug treatment for OxyContin abuse: a chart review of consecutive admissions to a substance abuse treatment facility in Kentucky. J Natl Compr Canc Netw 3:423–428
Jasinski DR, Martin WR (1967) Assessment of the dependence producing properties of dihydrocodeinone and codoxime. Clin Pharmacol Ther 8:266–270
Jasinski DR, Preston KL (1986) Comparison of intravenously administered methadone, morphine and heroin. Drug Alcohol Depend 17:301–310
Katz N, Fernandez K, Chang A, Benoit C, Butler SF (2008) Internet-based survey of nonmedical prescription opioid use in the United States. Clin J Pain 24:528–535
Lamb RJ, Preston KL, Schindler CW, Meisch RA, Katz DF, JL HJE, Goldberg SR (1991) The reinforcing and subjective effects of morphine in post-addicts: a dose-response study. J Pharmacol Exp Ther 259:1165–1173
Leow KP, Smith MT, Watt JA, Williams BE, Cramond T (1992) Comparative oxycodone pharmacokinetics in humans after intravenous, oral and rectal administration. Ther Drug Monit 14:479–484
Marsch LA, Bickel WK, Badger GJ, Rathmell JP, Swedberg MD, Jonzon B, Norsten-Höög C (2001) Effects of infusion rate of intravenously administered morphine on physiological, psychomotor, and self-reported measures in humans. J Pharmacol Exp Ther 299:1056–1065
Martin WR, Sloan BS, Sapira JD, Jasinski DR (1971) Physiologic, subjective and behavioral effects of amphetamine, methamphetamine, ephedrine, phenmetrazine and methylphenidate in man. Clin Pharmacol Ther 12:245–258
McLeod DR, Griffiths RR, Bigelow GE, Yingling J (1982) An automated version of the digit symbol substitution test (DSST). Behav Res Meth Instrum 14:463–466
Meert TF, Vermeirsch HA (2005) A preclinical comparison between different opioids: antinociceptive versus adverse effects. Pharmacol Biochem Behav 80:309–326
Rosenblum A, Parrino M, Schnoll SH, Fong C, Maxwell C, Cleland CM, Magura S, Haddox JD (2007) Prescription opioid abuse among enrollees into methadone maintenance treatment. Drug Alcohol Depend 90:64–71
Saarialho-Kere U, Mattila MJ, Seppala T (1989) Psychomotor, respiratory and neuroendocrinological effects of a μ-opioid receptor agonist (Oxycodone) in healthy volunteers. Pharmacol Toxicol 65:252–257
Stanski DR, Greenblatt DJ, Lowenstein E (1978) Kinetics of intravenous and intramuscular morphine. Clin Pharmacol Ther 24:52–59
Substance Abuse and Mental Health Services Administration (2007) The NSDUH Report: patterns and trends in nonmedical prescription pain reliever use: 2002–2005. Office of Applied Studies, Rockville
Substance Abuse and Mental Health Services Administration (2009) Results from the 2008 National Survey on Drug Use and Health: National Findings. Office of Applied Studies, Rockville, HHS Publication No SMA 09-4434
Tarkilla P, Tuominen M, Lindgren L (1997) Comparison of respiratory effects of tramadol and oxycodone. J Clin Anesth 9:582–585
Teoh SK, Mendelson JH, Mello NK, Kuehnle J, Sintavanarong P, Rhoades EM (1993) Acute interactions of buprenorphine with intravenous cocaine and morphine: an investigational new drug phase I safety evaluation. J Clin Psychopharmacol 13:87–99
Teoh SK, Mello NK, Mendelson JH, Kuehnle J, Gastfriend DR, Rhoades E, Sholar W (1994) Buprenorphine effects on morphine- and cocaine-induced subjective responses by drug-dependent men. J Clin Psychopharmacol 14:15–27
Trescot AM, Datta S, Lee M, Hansen H (2008) Opioid pharmacology. Pain Physician 11:S133–S153
Walker DJ, Zacny JP (1999) Subjective, psychomotor, and physiological effects of cumulative doses of opioid mu agonists in healthy volunteers. J Pharmacol Exp Ther 289:1454–1464
Walsh SL, Preston KL, Bigelow GE, Stitzer ML (1995) Acute administration of buprenorphine in humans: partial agonist and blockade effects. J Pharmacol Exp Ther 274:361–372
Walsh SL, Nuzzo PA, Lofwall MR, Holtman JR Jr (2008) The relative abuse liability of oral oxycodone, hydrocodone and hydromorphone assessed in prescription opioid abusers. Drug Alcohol Depend 98:191–202
Zacny JP (2003) Characterizing the subjective, psychomotor, and physiological effects of a hydrocodone combination product (Hycodan) in non-drug-abusing volunteers. Psychopharmacology 165:146–156
Zacny JP, Gutierrez S (2003) Characterizing the subjective, psychomotor, and physiological effects of oral oxycodone in non-drug-abusing volunteers. Psychopharmacology 170:242–254
Zacny JP, Gutierrez S (2008) Subjective, psychomotor, and physiological effects profile of hydrocodone/acetaminophen and oxycodone/acetaminophen combination products. Pain Med 9:433–443
Zacny JP, Lichtor SA (2008) Within-subject comparison of the psychopharmacological profiles of oral oxycodone and oral morphine in non-drug-abusing volunteers. Psychopharmacology 196:105–116
Zacny JP, Gutierrez S (2009) Within-subject comparison of the psychopharmacological profiles of oral hydrocodone and oxycodone combination products in non-drug-abusing volunteers. Drug Alcohol Depend 101:107–114
Zacny JP, Gutierrez S, Bolbolan SA (2005) Profiling the subjective, psychomotor, and physiological effects of a hydrocodone/acetaminophen product in recreational drug users. Drug Alcohol Depend 78:243–252
Acknowledgements
This research was supported by a grant from the National Institute on Drug Abuse (5R01DA016718-05) to Sharon L. Walsh and by the University of Kentucky CRDOC. The authors declare no conflicts of interest relevant to this project. The authors with to thank Lori Craig, Jessica DiCentes and Elizabeth Tammen for technical assistance and Stacy Miller, Todd McCoun, Pieter Steyn and Marie Thompson for medical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stoops, W.W., Hatton, K.W., Lofwall, M.R. et al. Intravenous oxycodone, hydrocodone, and morphine in recreational opioid users: abuse potential and relative potencies. Psychopharmacology 212, 193–203 (2010). https://doi.org/10.1007/s00213-010-1942-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-010-1942-4