Abstract
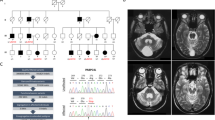
PINK1 gene mutations are a cause of recessively inherited, early-onset Parkinson’s disease. In some patients, a single heterozygous mutation has been identified, including the recurrent c.1366C>T transition. The interpretation of this finding remains controversial. Furthermore, the c.1366C>T mutation is associated with lower levels of PINK1 transcript, raising the question of whether mRNA levels correlate with the clinical status. We sequenced genomic DNA and copy DNA (cDNA) from 20 subjects carrying the c.1366C>T mutation in the homozygous (n = 5) or heterozygous (n = 15) state. In 17 mutation carriers, messenger RNA (mRNA) was quantified by real-time PCR using four different assays (PINK1 exon 5–6 or exon 7–8 relative to control genes SDHA or YWHAZ). Genomic sequencing confirmed the presence and zygosity of PINK1 mutations. cDNA sequencing in heterozygous mutation carriers revealed a strong wild-type and a much weaker or almost absent mutant signal, whereas in the homozygous patients, only the mutant signal was detected. Homozygous and heterozygous carriers showed PINK1 mRNA levels relative to a reference gene in the range of 0.1–0.2 and 0.5–0.6, respectively, compared with values of 0.9–1.0 in mutation-negative individuals. Treatment of lymphoblasts from a heterozygous mutation carrier with cycloheximide markedly increased the mutant transcript signal. We conclude that the recurrent PINK1 c.1366C>T mutation exerts a major effect at the mRNA level (80–90% reduction), most likely via nonsense-mediated mRNA decay. The absence of correlation between PINK1 mRNA levels and clinical status in heterozygous mutation carriers suggests that other genetic or environmental factors play a role in determining the phenotypic variability associated with the c.1366C>T mutation.




Similar content being viewed by others
References
Valente EM, Abou-Sleiman PM, Caputo V, Muqit MM, Harvey K, Gispert S, Ali Z, Del Turco D, Bentivoglio AR, Healy DG, Albanese A, Nussbaum R, Gonzalez-Maldonado R, Deller T, Salvi S, Cortelli P, Gilks WP, Latchman DS, Harvey RJ, Dallapiccola B, Auburger G, Wood NW (2004) Hereditary early-onset Parkinson’s disease caused by mutations in PINK1. Science 304:1158–1160
Valente EM, Salvi S, Ialongo T, Marongiu R, Elia AE, Caputo V, Romito L, Albanese A, Dallapiccola B, Bentivoglio AR (2004) PINK1 mutations are associated with sporadic early-onset parkinsonism. Ann Neurol 56:336–341
Bonifati V, Rohe CF, Breedveld GJ, Fabrizio E, De Mari M, Tassorelli C, Tavella A, Marconi R, Nicholl DJ, Chien HF, Fincati E, Abbruzzese G, Marini P, De Gaetano A, Horstink MW, Maat-Kievit JA, Sampaio C, Antonini A, Stocchi F, Montagna P, Toni V, Guidi M, Dalla Libera A, Tinazzi M, De Pandis F, Fabbrini G, Goldwurm S, de Klein A, Barbosa E, Lopiano L, Martignoni E, Lamberti P, Vanacore N, Meco G, Oostra BA (2005) Early-onset parkinsonism associated with PINK1 mutations: frequency, genotypes, and phenotypes. Neurology 65:87–95
Rogaeva E, Johnson J, Lang AE, Gulick C, Gwinn-Hardy K, Kawarai T, Sato C, Morgan A, Werner J, Nussbaum R, Petit A, Okun MS, McInerney A, Mandel R, Groen JL, Fernandez HH, Postuma R, Foote KD, Salehi-Rad S, Liang Y, Reimsnider S, Tandon A, Hardy J, St George-Hyslop P, Singleton AB (2004) Analysis of the PINK1 gene in a large cohort of cases with Parkinson disease. Arch Neurol 61:1898–1904
Klein C, Djarmati A, Hedrich K, Schafer N, Scaglione C, Marchese R, Kock N, Schule B, Hiller A, Lohnau T, Winkler S, Wiegers K, Hering R, Bauer P, Riess O, Abbruzzese G, Martinelli P, Pramstaller PP (2005) PINK1, Parkin, and DJ-1 mutations in Italian patients with early-onset parkinsonism. Eur J Hum Genet 13:1086–1093
Hedrich K, Hagenah J, Djarmati A, Hiller A, Lohnau T, Lasek K, Grunewald A, Hilker R, Steinlechner S, Boston H, Kock N, Schneider-Gold C, Kress W, Siebner H, Binkofski F, Lencer R, Munchau A, Klein C (2006) Clinical spectrum of homozygous and heterozygous PINK1 mutations in a large German family with Parkinson disease: role of a single hit? Arch Neurol 63:833–838
Klein C, Schlossmacher MG (2006) The genetics of Parkinson disease: implications for neurological care. Nat Clin Pract Neurol 2:136–146
Ibanez P, Lesage S, Lohmann E, Thobois S, De Michele G, Borg M, Agid Y, Durr A, Brice A (2006) Mutational analysis of the PINK1 gene in early-onset parkinsonism in Europe and North Africa. Brain 129:686–694
Zadikoff C, Rogaeva E, Djarmati A, Sato C, Salehi-Rad S, St George-Hyslop P, Klein C, Lang AE (2006) Homozygous and heterozygous PINK1 mutations: considerations for diagnosis and care of Parkinson’s disease patients. Mov Disord 21:875–879
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3:RESEARCH0034
Klein C, Grunewald A, Hedrich K (2006) Early-onset parkinsonism associated with PINK1 mutations: frequency, genotypes, and phenotypes. Neurology 66:1129–1130 (letter)
Abou-Sleiman PM, Muqit MM, McDonald NQ, Yang YX, Gandhi S, Healy DG, Harvey K, Harvey RJ, Deas E, Bhatia K, Quinn N, Lees A, Latchman DS, Wood NW (2006) A heterozygous effect for PINK1 mutations in Parkinson’s disease? Ann Neurol 60:414–419
Holbrook JA, Neu-Yilik G, Hentze MW, Kulozik AE (2004) Nonsense-mediated decay approaches the clinic. Nat Genet 36:801–808
Acknowledgment
This study was supported by grants from the Volkswagen Foundation, Germany (to C.K.) and from the Internationaal Parkinson Fonds, The Netherlands (to V.B.). We thank Ruud Koppenol, Erasmus MC, Rotterdam, for the artwork.
The experiments described in this paper comply with the current laws of the countries in which they were performed.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material:
Rights and permissions
About this article
Cite this article
Grünewald, A., Breedveld, G.J., Lohmann-Hedrich, K. et al. Biological effects of the PINK1 c.1366C>T mutation: implications in Parkinson disease pathogenesis. Neurogenetics 8, 103–109 (2007). https://doi.org/10.1007/s10048-006-0072-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10048-006-0072-y