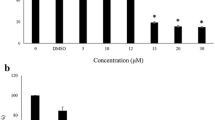
Abstract
Lower micromolar concentrations of peroxovanadium compound potassium bisperoxo(1,10-phenanthroline)oxovanadate (V) [bpV (phen)] stimulate RINm5F cell metabolic activity. 1 and 3 μmol/L bpV (phen) induces strong and sustained activation of extracellular signal-regulated kinase (ERK). However, it seems that bpV (phen) does not effect c-Jun N-terminal kinase (JNK) and p38 mitogen-activated protein kinase (MAPK) phosphorylation. In addition, bpV (phen) induces mitogen-activated protein kinase phosphatase-1 (MKP-1) expression. We found that ERK activation could be completely abolished if RINm5F cells were incubated with both bpV (phen) and PD 98059, a specific inhibitor of upstream ERK kinase MEK1. On the other hand, this combined treatment up-regulated activation of stress kinases, JNK and p38 MAPK, significantly suppressed MKP-1 expression and induced cell death. Thus, our results suggest that the mechanism underlying bpV (phen) survival-enhancing effect could be associated with induced ERK activation and MKP-1 expression.
Similar content being viewed by others
References
Bokemeyer D, Lindemann M, Kramer HJ. Regulation of mitogen-activated protein kinase phosphatase-1 in vascular smooth muscle cells. Hypertension. 1998;32:661–7.
Bokemeyer D, Sorokin A, Yan M, Ahn NG, Templeton DJ, Dunn MJ, Induction of mitogen-activated protein kinase phosphatase 1 by the stress-activated protein kinase signaling pathway but not by extracellular signal-regulated kinase in fibroblases. J Biol Chem. 1996;271:639–42.
Brichard SM, Ongemba LN, Henquin JC. Oral vanadate decreases muscle insulin resistance in obese fa/fa rats. Diabetologia. 1992;35:522–7.
Cerovac Z, Ban J, Morinville A, Yaccato K, Shaver A, Maysinger D. Activation of MAPK by potassium bisperoxo(1,10-phenanthroline)oxovanadate (V). Neurochem Int. 1999;34:337–44.
Chu Y, Solski PA, Khosravi-Far R, Der CJ, Kelly K. The mitogen-activated protein kinase phosphatases PAC1, MKP-1, and MKP-2 have unique substrate specificities and reduced activity in vivo towards the ERK2 sevenmaker mutation. J Biol Chem. 1996;271:6497–501.
Cruz TF, Morgan A, Min W. In vitro and vivo antineoplastic effects of ortovanadate. Mol Cell Biochem. 1995;153:161–6.
Daum G, Kalmes A, Levkau B, Wang V, Davies MG, Clowes AW. Pervanadate inhibits mitogen-activated protein kinase kinase-1 in a p38MAPK-dependent manner. FEBS Lett. 1998;427:271–4.
Dikic I, Schlessinger J, Lax I. PC12 cells overexpressing the insulin receptor undergo insulin-dependent neuronal differentiation. Curr Biol. 1994;4:702–8.
Dubyak GR, Kleinzeller AD. The insulin mimetic effects of vanadate in isolated rat adipocytes. J Biol Chem. 1980;255: 5306–12.
Franklin CC, Kraft AS. Conditional expression of the mitogen-activated protein kinase (MAPK) phosphatase MKP-1 preferentially inhibits p38 MAPK and stress-activated protein kinase in U937 cells. J Biol Chem. 1997;272:16917–23.
Franklin CC, Srikanth S, Kraft AS. Conditional expression of mitogen-activated protein kinase phosphatase-1, MKP-1, is cytoprotective against UV-induced apoptosis. Proc Natl Acad Sci USA. 1998;95:3014–19.
Gazdar AF, Chick WL, Oie HK, et al. Continuous, clonal, insulin-and somatostatin-secreting cell lines established from a transplantable rat islet cell tumor. Proc Natl Acad Sci USA. 1980;77:3519–23.
Grumont RJ, Rasko JE, Strasser A, Gerondakis S. Activation of the mitogen-activated protein kinase pathway induces transcription of the PAC-1 phosphatase gene. Mol Cell Biol. 1996;16:2913–21.
Haneda M, Sugimoto T, Kikkawa R. Mitogen-activated protein kinase phosphatase: a negative regulator of the mitogen-activated protein kinase cascade. Eur J Pharmacol. 1999;365:1–7.
Krady MM, Freyermuth S, Rogue P, Malviya AN. Pervanadate elicits proliferation and mediates activation of mitogen-activated protein (MAP) kinase in the nucleus. FEBS Lett. 1997;412:420–4.
Liu Y, Gorospe M, Holbrook NJ. Role of mitogen-activated protein kinase phosphatase during the cellular response to genotoxic stress. Inhibition of c-Jun N-terminal kinase activity and AP-1-dependent gene activation. J Biol Chem. 1995;270:8377–80.
Marshall CJ. Specificity of receptor tyrosine kinase signaling: transient versus sustained extracellular signal-regulated kinase activation. Cell. 1995;80:179–85.
Morinville A, Maysinger D, Shaver A. From Vanadis to Atropos: vanadium compounds as pharmacological tools in cell death signaling. Trends in Pharmacol Sci. 1998;19: 452–60.
Mustelin T, Brockdorff J, Gjörloff-Wingren A, et al. Lymphocyte activation: the coming of the protein tyrosine phosphatases. Front Biosci. 1998;3:1060–96.
Natarajan V, Scribner WM, Morris AJ, et al. Role of p38 MAP kinase in diperoxovanadate-induced phospholipase D activation in endothelial cells. Am J Physiol Lung Cell Mol Physiol. 2001;281:L435–49.
Posner BI, Faure R, Burgess JW, et al. Peroxovanadium compounds. A new class of potent phosphotyrosine phosphatase inhibitors which are insulin mimetics. J Biol Chem. 1994;269:4596–604.
Rogers MV, Buensuceso C, Montague F, Mahadevan L. Vanadate stimulates differentiation and neurite outgrowth in rat pheochromocytoma PC12 cells and neurite extension in human neuroblastoma SH-SY5Y cells. Neuroscience. 1994;60:479–94.
Rumora L, Barišić K, Maysinger D, Žanić-Grubišić T. BpV (phen) induces apoptosis of RINm5F cells by modulation of MAPKs and MKP-1. Biochem Biophys Res Commun. 2003;300:877–83.
Rumora L, Shaver A, Žanić Grubišić T, Maysinger D. MKP-1 as a target for pharmacological manipulations in PC12 cell survival. Neurochem Int. 2001;39:25–32.
Sekar N, Li J, Shechter Y. Vanadium salts as insulin substitutes: mechanisms of action, a scientific and therapeutic tool in diabetes mellitus research. Crit Rev Biochem Mol Biol. 1996;31:339–59.
Tolman EL, Barris E, Burns M, Pansini A, Partridge R. Effect of vanadium on glucose metabolism in vitro. Life Sci. 1979; 25:1159–64.
Traverse S, Seedorf K, Paterson H, Marshall CJ, Cohen P, Ullrich A. EGF triggers neuronal differentiation of PC12 cells that overexpress the EGF receptor. Curr Biol. 1994;4: 694–701
Wang H, Scott RE. Unique and selective mitogenic effects of vanadate on SV40-transformed cells. Mol Cell Biochem. 1995;153:59–67.
Xia Z, Dickens M, Raingeaud J, Davis RJ, Greenberg ME. Opposing effects of ERK and JNK-p38 MAP kinases on apoptosis. Science. 1995;270:1326–31.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rumora, L., Hadžija, M., Maysinger, D. et al. Positive regulation of ERK activation and MKP-1 expression by peroxovanadium complex bpV (phen). Cell Biol Toxicol 20, 293–301 (2004). https://doi.org/10.1007/s10565-004-5104-5
Issue Date:
DOI: https://doi.org/10.1007/s10565-004-5104-5