Abstract
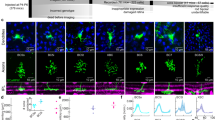
The mammalian visual system analyses the world through a set of separate spatio-temporal channels1,2. The organization of these channels begins in the retina1,3, where the precise laminations of both the axon terminals of bipolar cells and the dendritic arborizations of ganglion cells suggests the presence of a vertical stack of neural strata at the inner plexiform layer (IPL)3,4,5,6,7,8,9,10,11. Conversely, many inhibitory amacrine cell classes are multiply or diffusely stratified12, indicating that they might convey information between strata. On the basis of the diverse stratification and physiological properties of ganglion cells, it was suggested that the IPL contains a parallel set of representations of the visual world3,7 embodied in the strata and conveyed to higher centres by the classes of ganglion cells whose dendrites ramify at that stratum. Here we show that each stratum receives unique and substantively different excitatory and inhibitory neural inputs that are integrated to form at least ten different, parallel space-time spiking outputs. The response properties of these strata are ordered in the time domain. Inhibition through GABAC receptors extracts spatial edges in neural representations and seems to separate the functional properties of the strata. We describe a new form of neuronal interaction that we call ‘vertical inhibition’ that acts not laterally, but between strata.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Rodieck, R. W. The First Steps in Seeing (Sinauer, Sunderland, Massachusetts, 1998).
Albright, T. D., Jessell, T. M., Kandel, E. R. & Posner, M. I. Neural science: a century of progress and the mysteries that remain. Neuron 25, S1–S55 (2000).
Boycott, B. & Wässle, H. Parallel processing in the mammalian retina. The Proctor Lecture. Invest. Ophthalmol. Vis. Sci. 40, 1313–1328 (1999).
Euler, T. & Wässle, H. Immunocytochemical identification of cone bipolar cells in the rat retina. J. Comp. Neurol. 361, 461–478 (1995).
Euler, T., Schneider, H. & Wässle, H. Glutamate responses of bipolar cells in a slice preparation of the rat retina. J. Neurosci. 16, 2934–2944 (1996).
Cohen, E. & Sterling, P. Demonstration of cell types among cone bipolar neurons of cat retina. Phil. Trans. R. Soc. Lond. B 330, 305–321 (1990).
Maturana, H. R., Lettwin, J. Y., Pitts, W. H. & McCulloch, W. S. Physiology and anatomy of vision in the frog. J. Gen. Physiol. 43, 129–175 (1960).
Cajal, S. R. La rétine des vertebrés. La Cellule 9, 119–257 (1893).
Wu, S. M., Gao, F. & Maple, B. R. Functional architecture of synapses in the inner retina: segregation of visual signals by stratification of bipolar cell axon terminals. J. Neurosci. 20, 4462–4470 (2000).
Zhang, J., Li, W. & Massey, S. C. Cholinergic input to ganglion cells depends on depth in the IPL. Invest. Ophthalmol. 40, S812 (1999).
Famiglietti, E. V. Jr & Kolb, H. Structural basis for on- and off-center responses in retinal ganglion cells. Science 194, 193–195 (1976).
MacNeil, M. A. & Masland, R. H. Extreme diversity among amacrine cells: implications for function. Neuron 20, 971–982 (1998).
Roska, B., Nemeth, E., Orzo, L. & Werblin, F. S. Three levels of lateral inhibition: a space-time study of the retina of the tiger salamander. J. Neurosci. 20, 1941–1951 (2000).
Wässle, H. & Boycott, B. B. Functional architecture of the mammalian retina. Phys. Rev. 71, 447–480 (1991).
Fisher, S. K. & Boycott, B. B. Synaptic connections made by horizontal cells within the outer plexiform layer of the retina of the cat and the rabbit. Proc. R. Soc. Lond. B 186, 317–331 (1974).
Euler, T. & Wässle, H. Different contributions of GABAA and GABAC receptors to rod and cone bipolar cells in a rat retinal slice preparation. J. Neurophys. 79, 1384–1395 (1998).
Shields, C. R., Tran, M. N., Wong, R. O. & Lukasiewicz, P. D. Distinct ionotropic GABA receptors mediate presynaptic and postsynaptic inhibition in retinal bipolar cells. J. Neurosci. 20, 2673–2682 (2000).
Feigenspan, A., Wässle, H. & Bormann, J. Pharmacology of GABA receptor Cl- channels in rat retinal bipolar cells. Nature 361, 159–162 (1993).
Qian, H. & Dowling, J. E. Novel GABA responses from rod-driven retinal horizontal cells. Nature 361, 162–164 (1993).
Enz, R., Brandstätter, J. H., Wässle, H. & Bormann, J. Immunocytochemical localization of the GABAC receptor rho subunit in the mammalian retina. J. Neurosci. 16, 4479–4490 (1996).
Awatramani, G. B. & Slaughter, M. M. Origin of transient and sustained responses in ganglion cells of the retina. J. Neurosci. 20, 7087–7095 (2000).
Bloomfield, S. A. & Miller, R. F. A functional organization of ON and OFF pathways in the rabbit retina. J. Neurosci. 6, 1–13 (1986).
Slaughter, M. M. & Miller, R. F. 2-Amino-4-phosphonobutyric acid: a new pharmacological tool for retina research. Science 211, 182–185 (1981).
Wong, R. O. L. Retinal waves and visual system development. Annu. Rev. Neurosci. 22, 29–47 (1999).
Diamond, J. S. & Copenhagen, D. R. The relationship between light-evoked synaptic excitation and spiking behavior of salamander retinal ganglion cells. J. Physiol. 487, 711–725 (1995).
Amthor, F. R., Takahashi, E. S. & Oyster, C. W. Morphologies of rabbit retinal ganglion cells with concentric receptive fields. J. Comp. Neurol. 280, 72–96 (1989).
Acknowledgements
We thank J. E. Dowling, M. M. Poo, R. H. Kramer and S. Picaud for their comments on the manuscript, and C. Chen for his assistance with the experiments. This study was supported by the ONR and NIH.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Roska, B., Werblin, F. Vertical interactions across ten parallel, stacked representations in the mammalian retina. Nature 410, 583–587 (2001). https://doi.org/10.1038/35069068
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35069068
This article is cited by
-
Ancestral photoreceptor diversity as the basis of visual behaviour
Nature Ecology & Evolution (2024)
-
Birds multiplex spectral and temporal visual information via retinal On- and Off-channels
Nature Communications (2023)
-
Afactivism about understanding cognition
European Journal for Philosophy of Science (2023)
-
Glial Bmal1 role in mammalian retina daily changes
Scientific Reports (2022)
-
An epifluidic electronic patch with spiking sweat clearance for event-driven perspiration monitoring
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.