Abstract
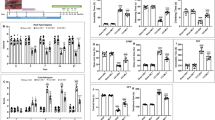
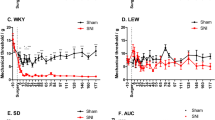
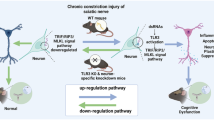
The physiological link between neuropathic pain and depression remains unknown despite a high comorbidity between these two disorders. A mouse model of spared nerve injury (SNI) was used to test the hypothesis that nerve injury precipitates depression through the induction of inflammation in the brain, and that prior exposure to stress exacerbates the behavioral and neuroinflammatory consequences of nerve injury. As compared with sham surgery, SNI induced mechanical allodynia, and significantly increased depressive-like behavior. Moreover, SNI animals displayed increased interleukin-1β (IL-1β) gene expression within the frontal cortex and concurrent increases in the expression of glial fibrillary acidic protein (GFAP) within the periaqueductal grey (PAG). Additionally, exposure to chronic restraint stress for 2 weeks before SNI exacerbated mechanical allodynia and depressive-like behavior, and resulted in an increase in IL-1β gene expression in the frontal cortex and brain-derived neurotrophic factor (BDNF) gene expression in PAG. Treatment with metyrapone (MET), a corticosteroid synthesis inhibitor, before stress eliminated deleterious effects of chronic stress on SNI. Finally, this study showed that interference with IL-1β signaling, through administration of IL-1 receptor antagonist (IL-1ra), ameliorated the effects of neuropathic pain on depressive-like behavior. Taken together, these data suggest that peripheral nerve injury leads to increased cytokine expression in the brain, which in turn, contributes to the development of depressive-like behavior. Furthermore, stress can facilitate the development of depressive-like behavior after nerve injury by promoting IL-1β expression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Wise TN, Fishbain DA, Holder-Perkins V . Painful physical symptoms in depression: a clinical challenge. Pain Med 2007; 8 (Suppl 2): S75–S82.
Strouse TB . The relationship between cytokines and pain/depression: a review and current status. Curr Pain Headache Rep 2007; 11: 98–103.
Jensen MP, Chodroff MJ, Dworkin RH . The impact of neuropathic pain on health-related quality of life: review and implications. Neurology 2007; 68: 1178–1182.
Gatchel RJT . D.C. Psychological Approaches to Pain Management: A Practitioner's Handbook, Guilford Press: New York, 1996.
Classification of Chronic Pain. Proceedings of the IASP Task Force on Taxonomy. IASP Press. Seattle, 1994.
Lesser H, Sharma U, LaMoreaux L, Poole RM . Pregabalin relieves symptoms of painful diabetic neuropathy: a randomized controlled trial. Neurology 2004; 63: 2104–2110.
Wilkie DJ, Huang HY, Reilly N, Cain KC . Nociceptive and neuropathic pain in patients with lung cancer: a comparison of pain quality descriptors. J Pain Symptom Manage 2001; 22: 899–910.
Urban MO, Gebhart GF . Supraspinal contributions to hyperalgesia. Proc Natl Acad Sci USA 1999; 96: 7687–7692.
Vanegas H, Schaible HG . Descending control of persistent pain: inhibitory or facilitatory? Brain Res Brain Res Rev 2004; 46: 295–309.
Gebhart GF . Descending modulation of pain. Neurosci Biobehav Rev 2004; 27: 729–737.
Neugebauer V, Li W, Bird GC, Han JS . The amygdala and persistent pain. Neuroscientist 2004; 10: 221–234.
Xu H, Wu LJ, Wang H, Zhang X, Vadakkan KI, Kim SS et al. Presynaptic and postsynaptic amplifications of neuropathic pain in the anterior cingulate cortex. J Neurosci 2008; 28: 7445–7453.
Apkarian AV, Lavarello S, Randolf A, Berra HH, Chialvo DR, Besedovsky HO et al. Expression of IL-1beta in supraspinal brain regions in rats with neuropathic pain. Neurosci Lett 2006; 407: 176–181.
Fields HL, Basbaum AI . Central nervous system mechanisms of pain modulation. In: Wall PD, Melzack R (eds), Textbook of Pain, 4th edn. Churchill Livingston: Edinburgh, 1999, pp 309–329
Rhudy JL, Williams AE, McCabe KM, Nguyen MA, Rambo P . Affective modulation of nociception at spinal and supraspinal levels. Psychophysiology 2005; 42: 579–587.
Villemure C, Bushnell MC . Cognitive modulation of pain: how do attention and emotion influence pain processing? Pain 2002; 95: 195–199.
Dworkin RH, Gitlin MJ . Clinical aspects of depression in chronic pain patients. Clin J Pain 1991; 7: 79–94.
Raison CL, Capuron L, Miller AH . Cytokines sing the blues: inflammation and the pathogenesis of depression. Trends Immunol 2006; 27: 24–31.
Watkins LR, Maier SF . Immune regulation of central nervous system functions: from sickness responses to pathological pain. J Intern Med 2005; 257: 139–155.
Caspi A, Sugden K, Moffitt TE, Taylor A, Craig IW, Harrington H et al. Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene. Science 2003; 301: 386–389.
McBeth J, Silman AJ, Gupta A, Chiu YH, Ray D, Morriss R et al. Moderation of psychosocial risk factors through dysfunction of the hypothalamic-pituitary-adrenal stress axis in the onset of chronic widespread musculoskeletal pain: findings of a population-based prospective cohort study. Arthritis Rheum 2007; 56: 360–371.
Wood PB . Stress and dopamine: implications for the pathophysiology of chronic widespread pain. Med Hypotheses 2004; 62: 420–424.
da Silva Torres IL, Cucco SN, Bassani M, Duarte MS, Silveira PP, Vasconcellos AP et al. Long-lasting delayed hyperalgesia after chronic restraint stress in rats-effect of morphine administration. Neurosci Res 2003; 45: 277–283.
Cerqueira JJ, Pego JM, Taipa R, Bessa JM, Almeida OF, Sousa N . Morphological correlates of corticosteroid-induced changes in prefrontal cortex-dependent behaviors. J Neurosci 2005; 25: 7792–7800.
Mitra R, Sapolsky RM . Acute corticosterone treatment is sufficient to induce anxiety and amygdaloid dendritic hypertrophy. Proc Natl Acad Sci USA 2008; 105: 5573–5578.
Bourquin AF, Suveges M, Pertin M, Gilliard N, Sardy S, Davison AC et al. Assessment and analysis of mechanical allodynia-like behavior induced by spared nerve injury (SNI) in the mouse. Pain 2006; 122, 14.e1–14.
Tal M, Bennett GJ . Extra-territorial pain in rats with a peripheral mononeuropathy: mechano-hyperalgesia and mechano-allodynia in the territory of an uninjured nerve. Pain 1994; 57: 375–382.
Decosterd I, Allchorne A, Woolf CJ . Differential analgesic sensitivity of two distinct neuropathic pain models. Anesth Analg 2004; 99: 457–463, table of contents.
Porsolt RD, Bertin A, Jalfre M . Behavioral despair in mice: a primary screening test for antidepressants. Arch Int Pharmacodyn Ther 1977; 229: 327–336.
Porsolt RD, Brossard G, Hautbois C, Roux S . Rodent models of depression: forced swimming and tail suspension behavioral despair tests in rats and mice. Curr Protoc Neurosci 2001 Chapter 8: Unit 8 10A.
Rotllant D, Armario A . A single dose of metyrapone caused long-term dysregulation of the hypothalamic-pituitary-adrenal axis in the rat. Neuroscience 2005; 130: 427–434.
Craft TK, DeVries AC . Role of IL-1 in poststroke depressive-like behavior in mice. Biol Psychiatry 2006; 60: 812–818.
Alexander JK, DeVries AC, Popovich PG . Stress effects on neuropathic pain. Annual Meeting of the Society for Neuroscience: Atlanta, GA, 2006.
Gillespie CF, Nemeroff CB . Hypercortisolemia and depression. Psychosom Med 2005; 67 (Suppl 1): S26–S28.
DeBattista C, Belanoff J . The use of mifepristone in the treatment of neuropsychiatric disorders. Trends Endocrinol Metab 2006; 17: 117–121.
Dinkel K, MacPherson A, Sapolsky RM . Novel glucocorticoid effects on acute inflammation in the CNS. J Neurochem 2003; 84: 705–716.
de Pablos RM, Villaran RF, Arguelles S, Herrera AJ, Venero JL, Ayala A et al. Stress increases vulnerability to inflammation in the rat prefrontal cortex. J Neurosci 2006; 26: 5709–5719.
MacPherson A, Dinkel K, Sapolsky R . Glucocorticoids worsen excitotoxin-induced expression of pro-inflammatory cytokines in hippocampal cultures. Exp Neurol 2005; 194: 376–383.
Guo W, Robbins MT, Wei F, Zou S, Dubner R, Ren K . Supraspinal brain-derived neurotrophic factor signaling: a novel mechanism for descending pain facilitation. J Neurosci 2006; 26: 126–137.
Ceccatelli S, Ernfors P, Villar MJ, Persson H, Hokfelt T . Expanded distribution of mRNA for nerve growth factor, brain-derived neurotrophic factor, and neurotrophin 3 in the rat brain after colchicine treatment. Proc Natl Acad Sci USA 1991; 88: 10352–10356.
Conner JM, Lauterborn JC, Yan Q, Gall CM, Varon S . Distribution of brain-derived neurotrophic factor (BDNF) protein and mRNA in the normal adult rat CNS: evidence for anterograde axonal transport. J Neurosci 1997; 17: 2295–2313.
Dantzer R, O’Connor JC, Freund GG, Johnson RW, Kelley KW . From inflammation to sickness and depression: when the immune system subjugates the brain. Nat Rev Neurosci 2008; 9: 46–56.
Wolf G, Gabay E, Tal M, Yirmiya R, Shavit Y . Genetic impairment of interleukin-1 signaling attenuates neuropathic pain, autotomy, and spontaneous ectopic neuronal activity, following nerve injury in mice. Pain 2006; 120: 315–324.
Scholz J, Woolf CJ . The neuropathic pain triad: neurons, immune cells and glia. Nat Neurosci 2007; 10: 1361–1368.
Sweitzer S, Martin D, DeLeo JA . Intrathecal interleukin-1 receptor antagonist in combination with soluble tumor necrosis factor receptor exhibits an anti-allodynic action in a rat model of neuropathic pain. Neuroscience 2001; 103: 529–539.
Oka T, Aou S, Hori T . Intracerebroventricular injection of interleukin-1 beta induces hyperalgesia in rats. Brain Res 1993; 624: 61–68.
Tonosaki Y, Nishiyama K, Roubos EW, Sugiura Y . alpha-Melanophore-stimulating hormone (alpha-MSH) antagonizes interleukin-1beta-induced hyperalgesia and Fos expression in the paraventricular and arcuate nucleus of the rat. Neuroendocrinology 2005; 81: 167–173.
Zhang RX, Li A, Liu B, Wang L, Ren K, Zhang H et al. IL-1ra alleviates inflammatory hyperalgesia through preventing phosphorylation of NMDA receptor NR-1 subunit in rats. Pain 2008; 135: 232–239.
Xie W, Luo S, Xuan H, Chou C, Song G, Lv R et al. Betamethasone affects cerebral expressions of NF-kappaB and cytokines that correlate with pain behavior in a rat model of neuropathy. Ann Clin Lab Sci 2006; 36: 39–46.
Wang Z, Wang J, Li X, Yuan Y, Fan G . Interleukin-1 beta of Red nucleus involved in the development of allodynia in spared nerve injury rats. Exp Brain Res 2008; 188: 379–384.
Schiepers OJ, Wichers MC, Maes M . Cytokines and major depression. Prog Neuropsychopharmacol Biol Psychiatry 2005; 29: 201–217.
Koo JW, Duman RS . IL-1beta is an essential mediator of the antineurogenic and anhedonic effects of stress. Proc Natl Acad Sci USA 2008; 105: 751–756.
Levine J, Barak Y, Chengappa KN, Rapoport A, Rebey M, Barak V . Cerebrospinal cytokine levels in patients with acute depression. Neuropsychobiology 1999; 40: 171–176.
Hayley S, Poulter MO, Merali Z, Anisman H . The pathogenesis of clinical depression: stressor- and cytokine-induced alterations of neuroplasticity. Neuroscience 2005; 135: 659–678.
Anisman H, Ravindran AV, Griffiths J, Merali Z . Endocrine and cytokine correlates of major depression and dysthymia with typical or atypical features. Mol Psychiatry 1999; 4: 182–188.
Thomas AJ, Davis S, Morris C, Jackson E, Harrison R, O’Brien JT . Increase in interleukin-1beta in late-life depression. Am J Psychiatry 2005; 162: 175–177.
Yu YW, Chen TJ, Hong CJ, Chen HM, Tsai SJ . Association study of the interleukin-1 beta (C-511T) genetic polymorphism with major depressive disorder, associated symptomatology, and antidepressant response. Neuropsychopharmacology 2003; 28: 1182–1185.
Tanebe K, Nishijo H, Muraguchi A, Ono T . Effects of chronic stress on hypothalamic lnterleukin-1beta, interleukin-2, and gonadotrophin-releasing hormone gene expression in ovariectomized rats. J Neuroendocrinol 2000; 12: 13–21.
Goshen I, Kreisel T, Ben-Menachem-Zidon O, Licht T, Weidenfeld J, Ben-Hur T et al. Brain interleukin-1 mediates chronic stress-induced depression in mice via adrenocortical activation and hippocampal neurogenesis suppression. Mol Psychiatry 2008; 13: 717–728.
Acknowledgements
We thank ZM Weil for technical support and assistance with data analysis. This work was supported by grants from the American Heart Association (predoctoral fellowship to KK), National Institute of Neurological Disorders and Stroke Behavioral Core Grant P30 NS045758 (to ACD), 5R01NR010806 SNI Grant (to ACD).
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Norman, G., Karelina, K., Zhang, N. et al. Stress and IL-1β contribute to the development of depressive-like behavior following peripheral nerve injury. Mol Psychiatry 15, 404–414 (2010). https://doi.org/10.1038/mp.2009.91
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2009.91
Keywords
This article is cited by
-
Autophagy dysfunction contributes to NLRP1 inflammasome-linked depressive-like behaviors in mice
Journal of Neuroinflammation (2024)
-
The soluble epoxide hydrolase inhibitor TPPU improves comorbidity of chronic pain and depression via the AHR and TSPO signaling
Journal of Translational Medicine (2023)
-
Adaptation of prelimbic cortex mediated by IL-6/STAT3/Acp5 pathway contributes to the comorbidity of neuropathic pain and depression in rats
Journal of Neuroinflammation (2022)
-
Differential neural processing of unpleasant sensory stimulation in patients with major depression
European Archives of Psychiatry and Clinical Neuroscience (2021)
-
The role of microglia in chronic pain and depression: innocent bystander or culprit?
Psychopharmacology (2021)