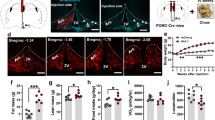
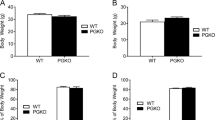
Abstract
Cholesterol circulates in the blood in association with triglycerides and other lipids, and elevated blood low-density lipoprotein cholesterol carries a risk for metabolic and cardiovascular disorders, whereas high-density lipoprotein (HDL) cholesterol in the blood is thought to be beneficial. Circulating cholesterol is the balance among dietary cholesterol absorption, hepatic synthesis and secretion, and the metabolism of lipoproteins by various tissues. We found that the CNS is also an important regulator of cholesterol in rodents. Inhibiting the brain's melanocortin system by pharmacological, genetic or endocrine mechanisms increased circulating HDL cholesterol by reducing its uptake by the liver independent of food intake or body weight. Our data suggest that a neural circuit in the brain is directly involved in the control of cholesterol metabolism by the liver.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Grundy, S.M. Metabolic syndrome: a multiplex cardiovascular risk factor. J. Clin. Endocrinol. Metab. 92, 399–404 (2007).
Cummings, D.E. Ghrelin and the short- and long-term regulation of appetite and body weight. Physiol. Behav. 89, 71–84 (2006).
Cowley, M.A. et al. The distribution and mechanism of action of ghrelin in the CNS demonstrates a novel hypothalamic circuit regulating energy homeostasis. Neuron 37, 649–661 (2003).
Cone, R.D. Studies on the physiological functions of the melanocortin system. Endocr. Rev. 27, 736–749 (2006).
Vaisse, C. et al. Melanocortin-4 receptor mutations are a frequent and heterogeneous cause of morbid obesity. J. Clin. Invest. 106, 253–262 (2000).
Ford, E.S., Giles, W.H. & Dietz, W.H. Prevalence of the metabolic syndrome among US adults: findings from the third National Health and Nutrition Examination Survey. J. Am. Med. Assoc. 287, 356–359 (2002).
Tschöp, M., Smiley, D.L. & Heiman, M.L. Ghrelin induces adiposity in rodents. Nature 407, 908–913 (2000).
Ma, X., Bruning, J. & Ashcroft, F.M. Glucagon-like peptide 1 stimulates hypothalamic proopiomelanocortin neurons. J. Neurosci. 27, 7125–7129 (2007).
Pocai, A., Obici, S., Schwartz, G.J. & Rossetti, L. A brain-liver circuit regulates glucose homeostasis. Cell Metab. 1, 53–61 (2005).
Sutton, G.M. et al. Diet-genotype interactions in the development of the obese, insulin-resistant phenotype of C57BL/6J mice lacking melanocortin-3 or -4 receptors. Endocrinology 147, 2183–2196 (2006).
Chen, A.S. et al. Inactivation of the mouse melanocortin-3 receptor results in increased fat mass and reduced lean body mass. Nat. Genet. 26, 97–102 (2000).
Schoonjans, K. et al. Liver receptor homolog 1 controls the expression of the scavenger receptor class B type I. EMBO Rep. 3, 1181–1187 (2002).
Lambert, G. et al. The farnesoid X-receptor is an essential regulator of cholesterol homeostasis. J. Biol. Chem. 278, 2563–2570 (2003).
Cettour-Rose, P. & Rohner-Jeanrenaud, F. The leptin-like effects of 3-d peripheral administration of a melanocortin agonist are more marked in genetically obese Zucker (fa/fa) than in lean rats. Endocrinology 143, 2277–2283 (2002).
Blüher, S. et al. Responsiveness to peripherally administered melanocortins in lean and obese mice. Diabetes 53, 82–90 (2004).
Enriori, P.J. et al. Diet-induced obesity causes severe but reversible leptin resistance in arcuate melanocortin neurons. Cell Metab. 5, 181–194 (2007).
Rigotti, A. et al. A targeted mutation in the murine gene encoding the high density lipoprotein (HDL) receptor scavenger receptor class B type I reveals its key role in HDL metabolism. Proc. Natl. Acad. Sci. USA 94, 12610–12615 (1997).
Varban, M.L. et al. Targeted mutation reveals a central role for SR-BI in hepatic selective uptake of high density lipoprotein cholesterol. Proc. Natl. Acad. Sci. USA 95, 4619–4624 (1998).
Tondu, A.L. et al. Insulin and angiotensin II induce the translocation of scavenger receptor class B, type I from intracellular sites to the plasma membrane of adipocytes. J. Biol. Chem. 280, 33536–33540 (2005).
Shetty, S., Eckhardt, E.R., Post, S.R. & van der Westhuyzen, D.R. Phosphatidylinositol-3-kinase regulates scavenger receptor class B type I subcellular localization and selective lipid uptake in hepatocytes. Arterioscler. Thromb. Vasc. Biol. 26, 2125–2131 (2006).
Ginsberg, H.N. Insulin resistance and cardiovascular disease. J. Clin. Invest. 106, 453–458 (2000).
Arai, T. et al. Increased plasma cholesteryl ester transfer protein in obese subjects. A possible mechanism for the reduction of serum HDL cholesterol levels in obesity. Arterioscler. Thromb. 14, 1129–1136 (1994).
Gruen, M.L. et al. Persistence of high density lipoprotein particles in obese mice lacking apolipoprotein A-I. J. Lipid Res. 46, 2007–2014 (2005).
Lundåsen, T., Liao, W., Angelin, B. & Rudling, M. Leptin induces the hepatic high density lipoprotein receptor scavenger receptor B type I (SR-BI) but not cholesterol 7alpha-hydroxylase (Cyp7a1) in leptin-deficient (ob/ob) mice. J. Biol. Chem. 278, 43224–43228 (2003).
Silver, D.L., Jiang, X.C. & Tall, A.R. Increased high density lipoprotein (HDL), defective hepatic catabolism of ApoA-I and ApoA-II, and decreased ApoA-I mRNA in ob/ob mice. Possible role of leptin in stimulation of HDL turnover. J. Biol. Chem. 274, 4140–4146 (1999).
Silver, D.L., Wang, N. & Tall, A.R. Defective HDL particle uptake in ob/ob hepatocytes causes decreased recycling, degradation, and selective lipid uptake. J. Clin. Invest. 105, 151–159 (2000).
Rader, D.J. Molecular regulation of HDL metabolism and function: implications for novel therapies. J. Clin. Invest. 116, 3090–3100 (2006).
Hasty, A.H. et al. Severe hypercholesterolemia, hypertriglyceridemia, and atherosclerosis in mice lacking both leptin and the low density lipoprotein receptor. J. Biol. Chem. 276, 37402–37408 (2001).
Huby, T. et al. Knockdown expression and hepatic deficiency reveal an atheroprotective role for SR-BI in liver and peripheral tissues. J. Clin. Invest. 116, 2767–2776 (2006).
Zhang, Y. et al. Hepatic expression of scavenger receptor class B type I (SR-BI) is a positive regulator of macrophage reverse cholesterol transport in vivo. J. Clin. Invest. 115, 2870–2874 (2005).
Engelking, L.J. et al. Schoenheimer effect explained—feedback regulation of cholesterol synthesis in mice mediated by Insig proteins. J. Clin. Invest. 115, 2489–2498 (2005).
Horton, J.D., Goldstein, J.L. & Brown, M.S. SREBPs: transcriptional mediators of lipid homeostasis. Cold Spring Harb. Symp. Quant. Biol. 67, 491–498 (2002).
Cummings, D.E. & Overduin, J. Gastrointestinal regulation of food intake. J. Clin. Invest. 117, 13–23 (2007).
Morton, G.J., Cummings, D.E., Baskin, D.G., Barsh, G.S. & Schwartz, M.W. Central nervous system control of food intake and body weight. Nature 443, 289–295 (2006).
Coll, A.P., Farooqi, I.S. & O′Rahilly, S. The hormonal control of food intake. Cell 129, 251–262 (2007).
Lowell, B.B. & Spiegelman, B.M. Towards a molecular understanding of adaptive thermogenesis. Nature 404, 652–660 (2000).
Ahrén, B. Autonomic regulation of islet hormone secretion—implications for health and disease. Diabetologia 43, 393–410 (2000).
Castañeda, T.R. et al. Obesity and the neuroendocrine control of energy homeostasis: the role of spontaneous locomotor activity. J. Nutr. 135, 1314–1319 (2005).
Lam, T.K. et al. Brain glucose metabolism controls the hepatic secretion of triglyceride-rich lipoproteins. Nat. Med. 13, 171–180 (2007).
Nogueiras, R. et al. The central melanocortin system directly controls peripheral lipid metabolism. J. Clin. Invest. 117, 3475–3488 (2007).
Schnölzer, M., Alewood, P., Jones, A., Alewood, D. & Kent, S.B. In situ neutralization in Boc-chemistry solid-phase peptide synthesis. Rapid, high yield assembly of difficult sequences. Int. J. Pept. Protein Res. 40, 180–193 (1992).
Pittman, R.C., Knecht, T.P., Rosenbaum, M.S. & Taylor, C.A. Jr. A nonendocytotic mechanism for the selective uptake of high-density lipoprotein–associated cholesterol esters. J. Biol. Chem. 262, 2443–2450 (1987).
Acknowledgements
This work was supported by US National Institute of Health grants NIDDK56863 (S.C.W. and M.H.T.), 5R01DK077975 (M.H.T.), R01 DK076907 (D.Y.H.) and HL67093 (W.S.D.). S.M.H. is the recipient of a Scientist Development Award from the American Heart Association (#0635079N). S.M.H. is a recipient of a Basic Science Award (1-10-BS-72) from the American Diabetes Association.
Author information
Authors and Affiliations
Contributions
D.P.-T., S.M.H., J.B., R.N. and P.T.P. designed and performed most of the experiments and wrote the manuscript. J.L.T. performed experiments, J.T.P. synthesized receptor ligands and A.A.B., S.C.B. and M.W.S. generated mouse models. N.G. carried out FPLC analyses. W.S.D. performed lipoprotein electrophoresis, E.G. and H.W.-P. carried out in vivo experiments, gene expression, inmunoblots and inmunoassays, M.A. performed surgical procedures and S.C.W. interpreted data and co-wrote the manuscript. R.D.D., D.Y.H. and M.H.T. conceptualized, analyzed and interpreted all studies and co-wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
R.D.D. and M.H.T. are scientific advisors for and shareholders of Marcadia Biotech and Ambrx Inc.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6, Supplementary Table 1 and Supplementary Methods (PDF 1132 kb)
Rights and permissions
About this article
Cite this article
Perez-Tilve, D., Hofmann, S., Basford, J. et al. Melanocortin signaling in the CNS directly regulates circulating cholesterol. Nat Neurosci 13, 877–882 (2010). https://doi.org/10.1038/nn.2569
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2569
This article is cited by
-
POMC neuronal heterogeneity in energy balance and beyond: an integrated view
Nature Metabolism (2021)
-
Brain transcriptome profile after CRISPR-induced ghrelin mutations in zebrafish
Fish Physiology and Biochemistry (2020)
-
Neuronal control of peripheral nutrient partitioning
Diabetologia (2020)
-
Melanocortin-4 receptor–regulated energy homeostasis
Nature Neuroscience (2016)
-
α-MSH and Foxc2 promote fatty acid oxidation through C/EBPβ negative transcription in mice adipose tissue
Scientific Reports (2016)