Abstract
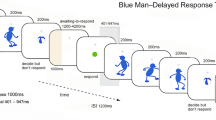
We investigate the hypothesis that those subregions of the prefrontal cortex (PFC) found to support proactive interference resolution may also support delay-spanning distractor interference resolution. Ten subjects performed delayed-recognition tasks requiring working memory for faces or shoes during functional MRI scanning. During the 15-sec delay interval, task-irrelevant distractors were presented. These distractors were either all faces or all shoes and were thus either congruent or incongruent with the domain of items in the working memory task. Delayed-recognition performance was slower and less accurate during congruent than during incongruent trials. Our fMRI analyses revealed significant delay interval activity for face and shoe working memory tasks within both dorsal and ventral PFC. However, only ventral PFC activity was modulated by distractor category, with greater activity for congruent than for incongruent trials. Importantly, this congruency effect was only present for correct trials. In addition to PFC, activity within the fusiform face area was investigated. During face distraction, activity was greater for face relative to shoe working memory. As in ventrolateral PFC, this congruency effect was only present for correct trials. These results suggest that the ventrolateral PFC and fusiform face area may work together to support delay-spanning interference resolution.
Article PDF
Similar content being viewed by others
References
Awh, E., Jonides, J., & Reuter-Lorenz, P. A. (1998). Rehearsal in spatial working memory. Journal of Experimental Psychology: Human Perception & Performance, 24, 780–790.
Awh, E., Jonides, J., Smith, E. E., Buxton, R. B., Frank, L. R., Love, T., Wong, E. C., & Gmeindl, L. (1999). Rehearsal in spatial working memory: Evidence from neuroimaging. Psychological Science, 10, 433–437.
Barnes, L., Nelson, J., & Reuter-Lorenz, P. (2001). Object-based attention and object working memory: Overlapping processes revealed by selective interference effects in humans. Progress in Brain Research, 134, 471–481.
Bunge, S. A., Ochsner, K. N., Desmond, J. E., Glover, G. H., & Gabrieli, J. D. E. (2001). Prefrontal regions involved in keeping information in and out of mind. Brain, 124, 2074–2086.
Cavada, C., & Goldman-Rakic, P. S. (1989). Posterior parietal cortex in rhesus monkey: II. Evidence for segregated corticocortical networks linking sensory and limbic areas with the frontal lobe. Journal of Cognitive Neuroscience, 287, 422–445.
Chafee, M. V., & Goldman-Rakic, P. S. (1998). Matching patterns of activity in primate prefrontal area 8a and parietal area 7ip neurons during a spatial working memory task. Journal of Neurophysiology, 79, 2919–2940.
Chafee, M. V., & Goldman-Rakic, P. S. (2000). Inactivation of parietal and prefrontal cortex reveals interdependence of neural activity during memory-guided saccades. Journal of Neurophysiology, 83, 1550–1566.
Chao, L. L., & Knight, R. T. (1995). Human prefrontal lesions increase distractibility to irrelevant sensory inputs. NeuroReport, 6, 1605–1610.
Chao, L. L., & Knight, R. T. (1998). Contribution of human prefrontal cortex to delay performance. Journal of Cognitive Neuroscience, 10, 167–177.
Constantinidis, C., & Steinmetz, M. (1996). Neural activity in posterior parietal area 7a during the delay periods of a spatial memory task. Journal of Neurophysiology, 76, 1352–1355.
Corbetta, M., Kincade, J. M., & Shulman, G. L. (2002). Neural systems for visual orienting and their relationships to spatial working memory. Journal of Cognitive Neuroscience, 14, 508–523.
Desimone, R., Chelazzi, L., Miller, E. K., & Duncan, J. (1995). Neuronal mechanisms of visual attention. In T. V. Papathomas (Ed.), Early vision and beyond (pp. 219–226). Cambridge, MA: MIT Press, Bradford Books.
Desimone, R., & Duncan, J. (1995). Neural mechanisms of selective visual attention. Annual Review of Neuroscience, 18, 193–222.
D’Esposito, M., Postle, B. R., Ballard, D., & Lease, J. (1999). Maintenance versus manipulation of information held in working memory: An event-related fMRI study. Brain & Cognition, 41, 66–86.
D’Esposito, M., Postle, B. R., Jonides, J., & Smith, E. E. (1999). The neural substrate and temporal dynamics of interference effects in working memory as revealed by event-related functional MRI. Proceedings of the National Academy of Sciences, 96, 7514–7519.
Epstein, R., & Kanwisher, N. (1998). A cortical representation of the local visual environment. Nature, 392, 598–601.
Friston, K. J., Ashburner, J., Frith, C. D., Poline, J.-B., Heather, J. D., & Frackowiak, R. S. J. (1995). Spatial registration and normalization of images. Human Brain Mapping, 2, 165–189.
Funahashi, S., Bruce, C. J., & Goldman-Rakic, P. S. (1989). Mnemonic coding of visual space in the monkey’s dorsolateral prefrontal cortex. Journal of Neurophysiology, 61, 331–349.
Fuster, J. M. (1973). Unit activity in prefrontal cortex during delayedresponse performance: Neuronal correlates of transient memory. Journal of Neurophysiology, 36, 61–78.
Fuster, J. M. (1995). Memory in the cerebral cortex: An empirical approach to neural networks in the human and nonhuman primate. Cambridge, MA: MIT Press.
Fuster, J. M. (1997). The prefrontal cortex: Anatomy, physiology, and neuropsychology of the frontal lobe (3rd ed.). Philadelphia: Lippincott-Raven.
Giesbrecht, B., & Mangun, G. R. (2002). The neural mechanisms of top-down control. In H.-O. Karnath, D. Milner, & G. Vallar (Eds.), The cognitive and neural bases of spatial neglect (pp. 243–257). Oxford: Oxford University Press.
Goggin, J., & Wickens, D. D. (1971). Proactive interference and language change in short-term memory. Journal of Verbal Learning & Verbal Behavior, 10, 453–458.
Goldman-Rakic, P. S. (1987). Circuitry of primate prefrontal cortex and regulation of behavior by representational memory. In J. M. Brookhart & V. B. Mountcastle (Series Eds.) & F. Plum (Vol. Ed.), Handbook of physiology: Sec. 1. The nervous system: Vol. 5. Higher functions of the brain (pp. 373–417). Bethesda, MD: American Physiological Society.
Hawkins, H. L., Pardo, V. J., & Cox, R. D. (1972). Proactive interference in short-term recognition: Trace interaction or competition? Journal of Experimental Psychology, 92, 43–48.
Hopfinger, J. B., Buonocore, M. H., & Mangun, G. R. (2000). The neural mechanisms of top-down attentional control. Nature Neuroscience, 3, 284–291.
Jha, A. P. (2002). Tracking the time-course of attentional involvement in spatial working memory: An event-related potential investigation. Cognitive Brain Research, 15, 61–69.
Jha, A. P., & McCarthy, G. (2000). The influence of memory load upon delay-interval activity in a working-memory task: An eventrelated functional MRI study. Journal of Cognitive Neuroscience, 12, 90–105.
Jiang, Y., Haxby, J. V., Martin, A., Ungerleider, L. G., & Parasuraman, R. (2000). Complementary neural mechanisms for tracking items in human working memory. Science, 287, 643–646.
Jonides, J., Smith, E. E., Marshuetz, C., Koeppe, R. A., & Reuter-Lorenz, P. A. (1998). Inhibition in verbal working memory revealed by brain activation. Proceedings of the National Academy of Sciences, 95, 8410–8413.
Kan, I. P., & Thompson-Schill, S. L. (2004). Effect of name agreement on prefrontal activity during overt and covert picture naming. Cognitive, Affective, & Behavioral Neuroscience, 4, 43–57.
Kanwisher, N., McDermott, J., & Chun, M. M. (1997). The fusiform face area: A module in human extrastriate cortex specialized for face perception. Journal of Neuroscience, 17, 4302–4311.
Kastner, S., Pinsk, M. A., De Weerd, P., Desimone, R., & Ungerleider, L. G. (1999). Increased activity in human visual cortex during directed attention in the absence of visual stimulation. Neuron, 22, 751–761.
Kimberg, D. Y., & Farah, M. J. (1993). A unified account of cognitive impairments following frontal lobe damage: The role of working memory in complex, organized behavior. Journal of Experimental Psychology: General, 122, 411–428.
Knight, R. T., Hillyard, S. A., Woods, D. L., & Neville, H. J. (1981). The effects of frontal cortex lesions on event-related potentials during auditory selective attention. Electroencephalography & Clinical Neurophysiology, 52, 571–582.
Knight, R. T., Staines, W. R., Swick, D., & Chao, L. L. (1999). Prefrontal cortex regulates inhibition and excitation in distributed neural networks. Acta Psychologica, 101, 159–178.
LaBar, K. S., Gitelman, D. R., Parrish, T. B., & Mesulam, M.-M. (1999). Neuroanatomic overlap of working memory and spatial attention networks: A functional MRI comparison within subjects. NeuroImage, 10, 695–704.
Leung, H.-C., Gore, J. C., & Goldman-Rakic, P. S. (2002). Sustained mnemonic response in the human middle frontal gyrus during on-line storage of spatial memoranda. Journal of Cognitive Neuroscience, 14, 659–671.
Luria, A. R. (1973). The working brain: An introduction to neuropsychology (B. Haigh, Trans.). Harmondsworth, U.K.: Penguin.
Luria, A. R., & Homskaya, E. D. (1970). Frontal lobes and the regulation of arousal processes. In D. I. Mostofsky (Ed.), Attention: Contemporary theory and analysis (pp. 58–72). New York: Appleton-Century-Crofts.
Malmo, R. (1942). Interference factors in delayed response in monkeys after removal of frontal lobes. Journal of Neurophysiology, 5, 295–308.
Mangun, G. R., Buonocore, M. H., Girelli, M., & Jha, A. P. (1998). ERP and fMRI measures of visual spatial selective attention. Human Brain Mapping, 6, 383–389.
Marcer, D. (1972). Interference and short-term retention. British Journal of Psychology, 63, 533–536.
May, C. P., Hasher, L., & Kane, M. J. (1999). The role of interference in memory span. Memory & Cognition, 27, 759–767.
McCarthy, G., Puce, A., Gore, J. C., & Allison, T. (1997). Facespecific processing in the human fusiform gyrus. Journal of Cognitive Neuroscience, 9, 605–610.
McIntyre, J. S., Stojak, R. A., & Mostoway, W. (1973). Individual organization and release from proactive interference. Journal of Experimental Psychology, 98, 164–168.
Milham, M. P., Banich, M. T., Webb, A., Barad, V., Cohen, N. J., Wszalek, T., & Kramer, A. F. (2001). The relative involvement of anterior cingulate and prefrontal cortex in attentional control depends on nature of conflict. Cognitive Brain Research, 12, 467–473.
Miller, E. K., & Cohen, J. D. (2001). An integrative theory of prefrontal cortex function.Annual Review of Neuroscience, 24, 167–202.
Miller, E. K., Erickson, C. A., & Desimone, R. (1996). Neural mechanisms of visual working memory in prefrontal cortex of the macaque. Journal of Neuroscience, 16, 5154–5167.
Miller, E. K., Li, L., & Desimone, R. (1991). A neural mechanism for working and recognition memory in inferior temporal cortex. Science, 254, 1377–1379.
Miller, E. K., Li, L., & Desimone, R. (1993). Activity of neurons in anterior inferior temporal cortex during a short-term memory task. Journal of Neuroscience, 13, 1460–1478.
Nelson, J. K., Reuter-Lorenz, P. A., Sylvester, C.-Y. C., Jonides, J., & Smith, E. E. (2003). Dissociable neural mechanisms underlying response-based and familiarity-based conflict in working memory. Proceedings of the National Academy of Sciences, 100, 11171–11175.
Pessoa, L., Kastner, S., & Ungerleider, L. G. (2003). Neuroimaging studies of attention: From modulation of sensory processing to topdown control. Journal of Neuroscience, 23, 3990–3998.
Postle, B. R., Druzgal, T. J., & D’Esposito, M. (2003). Seeking the neural substrates of working memory storage. Cortex, 39, 927–946.
Postle, B. R., Zarahn, E., & D’Esposito, M. (2000). Using event-related fMRI to assess delay-period activity during performance of spatial and nonspatial working memory tasks. Brain Research Protocols, 5, 57–66.
Ranganath, C., DeGutis, J., & D’Esposito, M. (2004). Categoryspecific modulation of inferior temporal activity during working memory encoding and maintenance. Cognitive Brain Research, 20, 37–45.
Rao, S. G., Williams, G. V., & Goldman-Rakic, P. S. (1999). Isodirectional tuning of adjacent interneurons and pyramidal cells during working memory: Evidence for microcolumnar organization in PFC. Journal of Neurophysiology, 81, 1903–1916.
Rypma, B., & D’Esposito, M. (2000). Isolating the neural mechanisms of age-related changes in human working memory. Nature Neuroscience, 3, 509–515.
Rypma, B., Prabhakaran, V., Desmond, J. E., Glover, G. H., & Gabrieli, J. D. E. (1999). Load-dependent roles of frontal brain regions in the maintenance of working memory. NeuroImage, 9, 216–226.
Sakai, K., Rowe, J. B., & Passingham, R. E. (2002). Active maintenance in prefrontal area 46 creates distractor-resistant memory. Nature Neuroscience, 5, 479–484.
Thompson-Schill, S. L. (2003). Neuroimaging studies of semantic memory: Inferring “how” from “where.” Neuropsychologia, 41, 280–292.
Thompson-Schill, S. L., D’Esposito, M., Aguirre, G. K., & Farah, M. J. (1997). Role of left inferior prefrontal cortex in retrieval of semantic knowledge: A reevaluation. Proceedings of the National Academy of Sciences, 94, 14792–14797.
Thompson-Schill, S. L., Jonides, J., Marshuetz, C., Smith, E. E., D’Esposito, M., Kan, I. P., Knight, R. T., & Swick, D. (2002). Effects of frontal lobe damage on interference effects in working memory. Cognitive, Affective, & Behavioral Neuroscience, 2, 109–120.
Tippett, L. J., Gendall, A., Farah, M. J., & Thompson-Schill, S. L. (2004). Selection ability in Alzheimer’s disease: Investigation of a component of semantic processing. Neuropsychology, 18, 163–173.
Wencil, E. B., Rauch, S., Scheinker, D., & Jha, A. P. (2003). Faces are not always special! Working memory studies of distractor intrusiveness [Abstract]. Cognitive Neuroscience Society Abstracts: 2003 Annual Meeting, F236.
Wilson, F. A. W., Ö Scalaidhe, S. P., & Goldman-Rakic, P. S. (1993). Dissociation of object and spatial processing domains in primate prefrontal cortex. Science, 260, 1955–1958.
Zarahn, E., Aguirre, G. [K.], & D’Esposito, M. (1997a). A trialbased experimental design for fMRI. NeuroImage, 6, 122–138.
Zarahn, E., Aguirre, G. K., & D’Esposito, M. (1997b). Empirical analyses of BOLD fMRI statistics. I. Spatially unsmoothed data collected under null-hypothesis conditions. NeuroImage, 5, 179–197.
Zarahn, E., Aguirre, G. K., & D’Esposito, M. (1999). Temporal isolation of the neural correlates of spatial mnemonic processing with fMRI. Cognitive Brain Research, 7, 255–268.
Author information
Authors and Affiliations
Corresponding author
Additional information
This research was supported by a grant from the University of Pennsylvania Research Foundation.
Rights and permissions
About this article
Cite this article
Jha, A.P., Fabian, S.A. & Aguirre, G.K. The role of prefrontal cortex in resolving distractor interference. Cognitive, Affective, & Behavioral Neuroscience 4, 517–527 (2004). https://doi.org/10.3758/CABN.4.4.517
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.3758/CABN.4.4.517