Summary
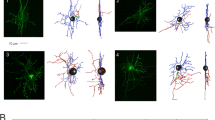
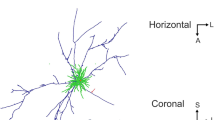
Stellate cells were studied in rapid Golgi preparations and in electron micrographs. These small neurons can be classified on the basis of their position in the molecular layer and the patterns of their dendritic and axonal arborizations as follows: (1) superficial cells with short, contorted dendrites and a circumscribed axonal arbor (upper third of the molecular layer); (2) deep stellate cells with radiating, twisted dendrites and with long axons giving rise to thin, varicose collaterals (middle third of the molecular layer); (3) deep stellate cells with similar dendrites and long axons giving collaterals to the basket around the Purkinje cell bodies (middle third of the molecular layer). An important characteristic of the stellate cell axon is that it generates most of its collaterals close to its origin. Even in long axon cells, only a few collaterals issue from the more distant parts of the axon. These forms contrast with the basket cell, which sends out long, straighter dendrites, and an extended axon that first emits branches at some distance from its origin. Furthermore, basket cell axon collaterals are usually stout in contrast to the frail, beaded collaterals of the stellate cell axon. The two cell types are considered to be distinct.
In electron micrographs stellate cells display folded nuclei and sparse cytoplasm with the characteristics usual for small neurons. Mitochondria are often the most conspicuous organelles because of their size and pleomorphism. The dendrites cannot be followed for long distances in thin sections because of their irregular caliber and course. Axons can be recognized on the basis of their appearance in Golgi preparations as short stretches of slender fibers distended at close intervals and running athwart the grid of the parallel fibers. These distensions, full of ovoid or flattened vesicles, synapse on the shafts of Purkinje cell dendrites and also on the dendrites of Golgi cells, basket cells, and other stellate cells. In all cases the synaptic complex occupies about a third of the junctional interface, the synaptic cleft is somewhat widened, and the pre- and postsynaptic dense plaques are thin and almost symmetrical.
Varicosities in the parallel fibers synapse with the soma and dendrites of stellate cells. These junctions display a widened synaptic cleft and asymmetrical pre- and postsynaptic densities. Junctions with climbing fibers (Scheibel collaterals) have also been seen.
The form of the stellate cell indicates that it plays a role in cerebellar circuitry different from that of the basket cell, although both cells are inhibitory. It is probably concerned with local effects on Purkinje cell dendrites within the field of its afferent parallel fibers.
Similar content being viewed by others
References
Anderson, P., Eccles, J. C., Voorhoeve, P. E.: Postsynaptic inhibition of cerebellar Purkinje cells. J. Neurophysiol. 27, 1138–1153 (1964).
Chan-Palay, V.: The recurrent collaterals of Purkinje cell axons: A correlated study of the rat's cerebellar cortex with electron microscopy and the Golgi method. Z. Anat. Entwickl.-Gesch. 134, 200–234 (1971).
Chan-Palay, V., Palay, S. L.: Interrelations of basket cell axons and climbing fibers in the cerebellar cortex of the rat. Z. Anat. Entwickl.-Gesch. 132, 191–227 (1970).
Eccles, J., Ito, M., Szentágothai, J.: The cerebellum as a neuronal machine. Berlin-Heidelberg-New York: Springer 1967.
Eccles, J. C., Llinás, R., Sasaki, K.: The inhibitory interneurones within the cerebellar cortex. Exp. Brain Res. 1, 1–16 (1966a).
Eccles, J. C., Llinás, R., Sasaki, K.: Parallel fibre stimulation and the responses induced thereby in the Purkinje cells of the cerebellum. Exp. Brain Res. 1, 17–39 (1966b).
Eccles, J., Llinás, R., Sasaki, K.: The action of antidromic impulses on the cerebellar Purkinje cells. J. Physiol. (Lond.) 182, 316–345 (1966c).
Eccles, J. C., Tábotiková, H., Tsukahara, N.: Responses of the Purkinje cells of a selachian cerebellum (Mustelus canis). Brain Res. 17, 57–86 (1970).
Fusari, R.: Sull origine delle fibre, nervose nello strato molecolare delle circonvoluzione cerebellari dell'uomo. Atti Reale Accad.-Sci. (Torino) 19, 47–51 (1883).
Gray, E. G.: Axosomatic and axo-dendritic synapses of the cerebral cortex: An electron microscopic study. J. Anat. (Lond.) 93, 420–433 (1959).
Jansen, J., Brodal, A.: Das Kleinhirn. Handbuch der Mikroskopischen, Anatomie des Menschen, Band IV/1 (Hrsg. W. Bargmann). Berlin-Göttingen-Heidelberg: Springer 1958.
Kitai, S. T., Shimono, T., Kennedy, D. T.: Inhibition in the cerebellar cortex of the lizard, Lacerta viridis. In: Neurobiology of cerebellar evolution and development (R. Llinás, ed.), p. 481–492. Chicago: AMA-ERF Institute for Biomedical Research 1969.
Larsell, O.: The morphogenesis and adult pattern of the lobules and fissures of the cerebellum of the white rat. J. comp. Neurol. 97, 281–356 (1952).
Lemkey-Johnston, N., Larramendi, L. M. H.: Morphological characteristics of mouse stellate basket cells and their neurological envelope: An electron microscopic study. J. comp. Neurol. 134, 39–72 (1968a).
Lemkey-Johnston, N., Larramendi, L. M. H.: Types and distribution of synapses upon basket and stellate cells of the mouse cerebellum. J. comp. Neurol. 134, 73–112 (1968b).
Llinás, R., Nicholson, C.: Electrophysiological analysis of alligator cerebellar cortex. A study on dendritic spikes. In: Neurobiology of cerebellar evolution and development (R. Llinás, ed.), p. 431–466. Chicago: AMA-ERF Institute for Biomedical Research 1969.
Nicholson, C., Llinás, R., Precht, W.: Neural elements of the cerebellum in elasmobranch fishes: Structural and functional characteristics. In: Neurobiology of cerebellar evolution and development (R. Llinás, ed.), p. 215–243. Chicago: AMA-ERF Institute for Biomedical Research 1969.
Palkovits, M., Magyar, P., Szentágothai, J.: Quantitative histological analysis of the cerebellar cortex in the cat. III. Structural organization of the molecular layer. Brain Res. 34, 1–18 (1971).
Ponti, U.: Sulla corteccia cerebellare della Cavia. Monit. zool. ital. 8, 36–40 (1897).
Rakic, P.: Extrinsic cytological determinants of basket and stellate cell dendritic pattern in the cerebellar molecular layer. J. comp. Neurol. In press.
Ramón y Cajal, S.: Sur l'origine et la direction des prolongations nerveuses de la couche moléculaire du cervelet. Int. Mschr. Anat. Physiol. 6, 158–174 (1889).
Ramón y Cajal, S.: Histologie du système nerveux de l'homme et des vertébrés (trans. by L. Azoulay), vol. 2. Paris: Maloine 1911.
Rushmer, D. S., Woodward, D. J.: Inhibition of Purkinje cells in the frog cerebellum. I. Evidence for a stellate cell inhibitory pathway. Brain Res. 33, 83–90 (1971).
Scheibel, M. E., Scheibel, A. B.: Observations on the intracortical relations of the climbing fibers of the cerebellum. J. comp. Neurol. 101, 733–763 (1954).
Smirnow, A. E.: Über eine besondere Art von Nervenzellen der Molecularschicht des Kleinhirns bei erwachsenen Säugetieren und beim Menschen. Anat. Anz. 13, 636–642 (1897).
Sotelo, C.: Ultrastructural aspects of the cerebellar cortex of the frog. In: Neurobiology of cerebellar evolution and development (R. Llinás, ed.), p. 327–371. Chicago: AMA-ERF Institute for Biomedical Research 1969.
Sotelo, C.: Stellate cells and their synapses on Purkinje cells in the cerebellum of the frog. Brain Res. 17, 510–514 (1970).
Author information
Authors and Affiliations
Additional information
Supported by U. S. Public Health Service Research Grant NS03659 and Training Grant NS05591 from the National Institute of Neurological Diseases and Stroke.
Rights and permissions
About this article
Cite this article
Chan-Palay, V., Palay, S.L. The stellate cells of the rat's cerebellar cortex. Z. Anat. Entwickl. Gesch. 136, 224–248 (1972). https://doi.org/10.1007/BF00519180
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00519180