Abstract
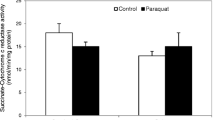
The effects of paraquat on rat brain were studied. Activities of complex I (NADH: ubiquinone oxidoreductase) in mitochondrial electron transport system, lipid peroxidation and the amount of catecholamines in rat brain were measured after acute paraquat exposure. Complex I activities were significantly lower and lipid peroxides were higher in the brains of a paraquat-treated group than in those of a control group. Lipid peroxide in rat serum, however, did not increase after paraquat exposure. A study of the time dependency of paraquat effects disclosed that mitochondrial complex I activities in rat brain as well as those in rat lung and liver gradually decreased prior to the appearance of respiratory dysfunction. As compared to controls, the dopamine in rat striatum was significantly lower in the paraquat-treated group. These results suggest that paraquat after crossing the blood-brain barrier might be reduced to the radical in rat brain, which may damage the brain tissue, especially dopaminergic neurons in striatum. We therefore propose that cerebral damage should be taken into consideration on paraquat exposure. Patients may therefore need to be followed up after exposure to high doses of paraquat.
Similar content being viewed by others
References
Barbeau A, Dallaire L, Buu NT, Poirier J, Rucinska E (1985) Comparative behavioral, biochemical and pigmentary effects of MPTP, MPP+ and paraquat in rana pipiens. Life Sci 37: 1529–1538
Bindoff LA, Birch-Machin M, Cartlidge NEF, Parker WD, Turnbull DM (1989) Mitochondrial function in Parkinson’s disease. Lancet 2: 49
Blaszczyński M, Litwińska J, Zaborowska D, Biliński T (1985) The role of respiratory chain in paraquat toxicity in yeast. Acta Microbiol Polon 34: 243–254
Bus JS, Aust SD, Gibson JE (1974) Superoxide- and singlet oxygen-catalyzed lipid peroxidation as a possible mechanism for paraquat (methyl viologen) toxicity. Biochem Biophys Res Commun 58 [3]: 749–755
Clejan L, Cederbaum AI (1989) Synergistic interactions between NADPH-cytochrome P-450 reductase, paraquat, and iron in the generation of active oxygen radicals. Biochem Pharmacol 38 [11]: 1779–1786
Corasaniti MT, Strongoli MC, Nistico G (1990) Determination of paraquat in rat brain using ion-pair solid-phase extraction and reversed-phase high-performance liquid chromatography with ultraviolet detection. J Chromatogr 527: 189–195
Fukushima T, Yamada K, Hojo N, Isobe A, Shiwaku K, Yamane Y (1993) Mechanism of cytotoxicity of paraquat I: NADH oxidation and paraquat radical formation via complex I. Exp Toxicol Pathol 45: 345–349
Fukushima T, Yamada K, Hojo N, Isobe A, Shiwaku K, Yamane Y (1994) Mechanism of cytotoxicity of paraquat III: The effects of acute paraquat exposure on the electron transport system in rat mitochondria. Exp Toxicol Pathol 46: 437–441
Glowinski J, Iversen LL (1966) Regional studies of catecholamines in rat brain—I. The disposition of [3H] norepinephrine, [3H] dopamine, [3H] DOPA in various regions of the brain. J Neurochem 13: 655–669
Gornall AG, Bardawill CS, David MN (1949) Determination of serum proteins by means of the biuret reaction. J Biol Chem 177: 751–766
Grant HC, Lantos PL, Parkinson C (1980) Cerebral damage in paraquat poisoning. Histopathology 4: 185–195
Grcěvić N, Jadro-Šantel D, Jukić S (1977) Cerebral changes in paraquat poisoning. In: Neurotoxicology. Raven Press, New York, pp 469–484
Hirai K, Witschi H, Cote MG (1985) Mitochondrial injury of pulmonary alveolar epithelial cells in acute paraquat intoxication. Exp Mol Pathol 43: 242–252
Hughes JT (1988) Brain damage due to paraquat poisoning: a fatal case with neuropathological examination of the brain. Neurotoxicology 9: 243–248
Ikarashi Y, Maruyama Y, Stavinoha WB (1984) Study of the use of the microwave magnetic field for the rapid inactivation of brain enzymes. Jpn J Pharmacol 35: 371–387
Kato R, Iwasaki K, Noguchi H (1976) Stimulatory effect of FMN and methyl viologen on cytochrome P-450 dependent reduction of tertiary amine N-oxide. Biochem Biophys Res Commun 72 [1]: 267–274
Koller WC (1986) Paraquat and Parkinson’s disease. Neurology 36: 1147
Markey SP, Weisz A, Bacon JP (1986) Reduced paraquat does not exhibit MPTP-like neurotoxicity. J Anal Toxicol 10: 256
Mitsui A, Nohta H, Ohkura Y (1985) High-performance liquid chromatography of plasma catecholamines using 1,2-diphenylethylenediamine as pre-column fluorescence derivatization reagent. J Chromatogr 344: 61–70
Mizuno Y, Ohta S, Tanaka M, Takamiya S, Suzuki K, Sato T, Oya H, Ozawa T, Kagawa Y (1989) Deficiencies in complex I subunits of the respiratory chain in Parkinson’s disease. Biochem Biophys Res Commun 163: 1450–1455
Mukada T, Sasano N, Sato K (1978) Autopsy findings in a case of acute paraquat poisoning with extensive cerebral purpura. Tohoku J Exp Med 125: 253–263
Nagao M, Takatori T, Wu B, Terazawa K, Gotouda H, Akabane H (1991) Immunohistochemical localization of paraquat in lung and brain. Med Sci Law 31 [1]: 61–64
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95: 351–358
Parker WD, Boyson SJ, Parks JK (1989) Abnormalities of the electron transport chain in idiopathic Parkinson’s disease. Ann Neurol 26: 719–723
Rose MS, Lock EA, Smith LL, Wyatt I (1976) Paraquat accumulation: tissue and species specificity. Biochem Pharmacol 25: 419–423
Singer TP, Ramsay RR (1990) Mechanism of the neurotoxicity of MPTP. An update. FEBS Lett 274: 1–8
Thakar JH, Hassan MN (1988) Effects of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), cyperquat (MPP+) and paraquat on isolated mitochondria from rat striatum, cortex and liver. Life Sci 43: 143–149
Tomita M (1991) Comparison of one-electron reduction activity against the bipyridylium herbicides, paraquat and diquat, in microsomal and mitochondrial fractions of liver, lung and kidney (in vitro). Biochem Pharmacol 42: 303–309
Turrens JF, Boveris A (1980) Generation of superoxide anion by the NADH dehydrogenase of bovine heart mitochondria. Biochem J 191: 421–427
Yamada K, Fukushima T (1993) Mechanism of cytotoxicity of paraquat II: organ specificity of paraquat-stimulated lipid peroxidation in the inner membrane of mitochondria. Exp Toxicol Pathol 45: 375–380
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tawara, T., Fukushima, T., Hojo, N. et al. Effects of paraquat on mitochondrial electron transport system and catecholamine contents in rat brain. Arch Toxicol 70, 585–589 (1996). https://doi.org/10.1007/s002040050316
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002040050316