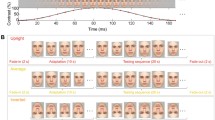
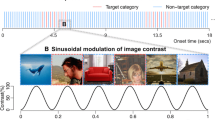
Abstract
This paper reviews a fast periodic visual stimulation (FPVS) approach developed recently to make significant progress in understanding visual discrimination of individual faces. Displaying pictures of faces at a periodic frequency rate leads to a high signal-to-noise ratio (SNR) response in the human electroencephalogram, at the exact frequency of stimulation, a so-called steady-state visual evoked potential (SSVEP, Regan in Electroencephalogr Clin Neurophysiol 20:238–248, 1966). For fast periodic frequency rates, i.e., between 3 and 9 Hz, this response is reduced if the exact same face identity is repeated compared to the presentation of different face identities, the largest difference being observed over the right occipito-temporal cortex. A 6-Hz stimulation rate (cycle duration of ~170 ms) provides the largest difference between different and repeated faces, as also evidenced in face-selective areas of the ventral occipito-temporal cortex in functional magnetic resonance imaging. This high-level discrimination response is reduced following inversion and contrast-reversal of the faces and can be isolated without subtraction thanks to a fast periodic oddball paradigm. Overall, FPVS provides a response that is objective (i.e., at an experimentally defined frequency), implicit, has a high SNR and is directly quantifiable in a short amount of time. Although the approach is particularly appealing for understanding face perception, it can be generalized to study visual discrimination of complex visual patterns such as objects and visual scenes. The advantages of the approach make it also particularly well-suited to investigate these functions in populations who cannot provide overt behavioral responses and can only be tested for short durations, such as infants, young children and clinical populations.















Similar content being viewed by others
Notes
In some SSVEP studies of low-level visual stimuli, an attribute of the stimulus can also change periodically, although the changes are limited to two states or smaller increasing or decreasing steps. For instance, a leftward motion alternates periodically with a rightward motion (Tyler and Kaitz 1977), or the phase of a sinusoidal grating reverses periodically (e.g., Atkinson et al. 1979). In the sweep SSVEP paradigm (Regan 1973), the same stimulus (e.g., a checkerboard) is presented at a periodic rate, but it increases or decreases progressively at every cycle according to a low-level property such as contrast, spatial frequency or orientation (e.g., Norcia and Tyler 1985; see Almoqbel et al. 2008 for a review).
The present approach should also be distinguished from the visually evoked spread spectrum response potential (VESPA, Lalor et al. 2006), in which the dimension of interest (usually luminance) changes according to a large number of known values, at fast rates (i.e., up to half the periodic rate of the stimulation monitor, usually 60 Hz). For luminance changes at least, this latter approach also provides a higher SNR than standard ERP measures (Lalor et al. 2006), although not in proportions that are comparable to the SNR obtained during periodic visual stimulation. However, by definition, the VESPA stimulation is not periodic, and the analysis is performed in the time-domain to extract the visual system’s impulse response, not in the frequency domain. The response of interest requires a strict modelization of the change of the property of interest (i.e., the stimulus luminance waveform), and to my knowledge, has not been applied to high-level visual properties (see Lalor et al. 2006, 2012).
References
Abbott LF, Rolls ET, Tovee MJ (1996) Representational capacity of face coding in monkeys. Cereb Cortex 6:498–505
Almoqbel F, Leat SJ, Irving E (2008) The technique, validity and clinical use of the sweep VEP. Ophthalmic Physiol Opt 28:393–403
Alonso-Prieto E, Belle G, Liu-Shuang J, Norcia AM, Rossion B (2013) The 6 Hz fundamental stimulation frequency rate for individual face discrimination in the right occipito-temporal cortex. Neuropsychologia 51:2863–2875
Appelbaum LG, Wade AR, Vildavski VY, Pettet MW, Norcia AM (2006) Cue-invariant networks for figure and background processing in human visual cortex. J Neurosci 26:11695–11708
Appelbaum LG, Ales JM, Cottereau B, Norcia AM (2010) Configural specificity of the lateral occipital cortex. Neuropsychologia 48:3323–3328
Atkinson J, Braddick O, French J (1979) Contrast sensitivity of the human neonate measured by the visual evoked potential. Invest Ophthalmol Vis Sci 18:210–213
Barton JJ (2008) Structure and function in acquired prosopagnosia: lessons from a series of 10 patients with brain damage. J Neuropsychol 2:197–225
Baylis GC, Rolls ET (1987) Responses of neurons in the inferior temporal cortex in short term and serial recognition memory tasks. Exp Brain Res 65:614–622
Behrmann M, Avidan G (2005) Congenital prosopagnosia: face-blind from birth. Trends Cogn Sci 9:180–187
Bentin S, McCarthy G, Perez E, Puce A, Allison T (1996) Electrophysiological studies of face perception in humans. J Cogn Neurosci 8:551–565
Benton AL, Van Allen MW (1968) Impairment in facial recognition in patients with cerebral disease. Trans Am Neurol Assoc 93:38–42
Bohórquez J, Ozdamar O, Açikgöz N, Yavuz E (2007) Methodology to estimate the transient evoked responses for the generation of steady state responses. Conf Proc IEEE Eng Med Biol Soc 2007:2444–2447
Bowles DC, McKone E, Dawel A, Duchaine B, Palermo R, Schmalzl L, Rivolta D, Wilson CE, Yovel G (2009) Diagnosing prosopagnosia: effects of ageing, sex, and participant-stimulus ethnic match on the Cambridge face memory test and Cambridge face perception test. Cogn Neuropsychol 26:423–455
Braddick OJ, Wattam-Bell J, Atkinson J (1986) Orientation-specific cortical responses develop in early infancy. Nature 320:617–619
Bruce V, Young A (1998) In the eye of the beholder: the science of face perception. Oxford University Press, Oxford
Busch NA, VanRullen R (2010) Spontaneous EEG oscillations reveal periodic sampling of visual attention. Proc Natl Acad Sci USA 107:16048–16053
Busigny T, Graf M, Mayer E, Rossion B (2010) Acquired prosopagnosia as a face-specific disorder: ruling out the general visual similarity account. Neuropsychologia 48:2051–2067
Buzsaki G, Llinas R, Singer W, Berthoz A, Christen Y (1994) Oscillatory and intermittent synchrony in the hippocampus: relevance to memory trace formation. In: Buzsaki G, Llinas R, Singer W, Berthoz A, Christen Y (eds) Temporal coding in the brain. Springer, Berlin, pp 145–172
Caharel S, d'Arripe O, Ramon M, Jacques C, Rossion B (2009) Early adaptation to unfamiliar faces across viewpoint changes in the right hemisphere: evidence from the N170 ERP component. Neuropsychologia 47:639–643
Capilla A, Pazo-Alvarez P, Darriba A, Campo P, Gross J (2011) Steady-state visual evoked potentials can be explained by temporal superposition of transient event-related responses. PLoS ONE 6:e14543
Carey S (1992) Becoming a face expert. Philos Trans R Soc Lond B Biol Sci 335(1273):95–102
Chen Y, Seth AK, Gally J, Edelman GM (2003) The power of human brain magnetoencephalographic signals can be modulated up or down by changes in an attentive visual task. Proc Natl Acad Sci USA 100:3501–3506
Crookes K, McKone E (2009) Early maturity of face recognition: no childhood development of holistic processing, novel face encoding, or face-space. Cognition 111:219–247
Davies-Thompson J, Gouws A, Andrews TJ (2009) An image-independent representation of familiar and unfamiliar faces in the human ventral stream. Neuropsychologia 47:1627–1635
de Heering A, Maurer D (2014) Face memory deficits in patients deprived of early visual input by bilateral congenital cataracts. Dev Psychobiol 56:96–108
Di Russo F, Pitzalis S, Aprile T, Spitoni G, Patria F, Stella A, Spinelli D, Hillyard SA (2007) Spatiotemporal analysis of the cortical sources of the steady-state visual evoked potential. Hum Brain Mapp 28:323–334
Ding J, Sperling G, Srinivasan R (2006) Attentional modulation of SSVEP power depends on the network tagged by the flicker frequency. Cereb Cortex 16:1016–1029
Duchaine BC, Nakayama K (2006a) The Cambridge face memory test: results for neurologically intact individuals and an investigation of its validity using inverted face stimuli and prosopagnosic participants. Neuropsychologia 44:576–585
Duchaine BC, Nakayama K (2006b) Developmental prosopagnosia: a window to content-specific face processing. Curr Opin Neurobiol 16:166–173
Freire A, Lee K, Symons LA (2000) The face-inversion effect as a deficit in the encoding of configural information: direct evidence. Perception 29:159–170
Galambos R, Makeig S, Talmachoff PJ (1981) A 40-Hz auditory potential recorded from the human scalp. Proc Natl Acad Sci USA 78:2643–2647
Galper RE (1970) Recognition of faces in photographic negative. Psychon Sci 19:207–208
Gauthier I, Tarr MJ, Moylan J, Skudlarski P, Gore JC, Anderson AW (2000) The FFA is part of a network that processes faces on an individual level. J Cogn Neurosci 12:495–504
Gentile F, Rossion B (2014) Temporal frequency tuning of cortical face-sensitive areas for individual face perception. NeuroImage 90:256–265
George N, Evans J, Fiori N, Davidoff J, Renault B (1996) Brain events related to normal and moderately scrambled faces. Cogn Brain Res 4:65–76
Gerlicher AM, van Loon AM, Scholte HS, Lamme VA, van der Leij AR (2014) Emotional facial expressions reduce neural adaptation to face identity. Soc Cogn Affect Neurosci. doi:10.1093/scan/nst022
Germine LT, Duchaine B, Nakayama K (2011) Where cognitive development and aging meet: face learning ability peaks after age 30. Cognition 118:201–210
Grill-Spector K, Malach R (2001) fMR-adaptation: a tool for studying the functional properties of human cortical neurons. Acta Psychol (Amst) 107(1–3):293–321
Grill-Spector K, Henson R, Martin A (2006) Repetition and the brain: neural models of stimulus-specific effects. Trends Cogn Sci 10(1):14–23
Gruss LF, Wieser MJ, Schweinberger SR, Keil A (2012) Face-evoked steady-state visual potentials: effects of presentation rate and face inversion. Front Hum Neurosci 6:316
Haig ND (1985) How faces differ—a new comparative technique. Perception 14:601–615
Heinrich SP (2010) Some thoughts on the interpretation of steady-state evoked potentials. Doc Ophthalmol 120:205–214
Heinrich SP, Mell D, Bach M (2009) Frequency-domain analysis of fast oddball responses to visual stimuli: a feasibility study. Int J Psychophysiol 73:287–293
Henson RN (2003) Neuroimaging studies of priming. Prog Neurobiol 70:53–81
Itier RJ, Taylor MJ (2002) Inversion and contrast polarity reversal affect both encoding and recognition processes of unfamiliar faces: a repetition study using ERPs. Neuroimage 15:353–372
Jacques C, Rossion B (2006) The speed of individual face categorization. Psychol Sci 17:485–492
Jacques C, d’Arripe O, Rossion B (2007) The time course of the inversion effect during individual face discrimination. J Vis 7:1–9
Jeffreys DA (1989) A face-responsive potential recorded from the human scalp. Exp Brain Res 78:193–202
Jeffreys DA (1993) The influence of stimulus orientation on the vertex positive scalp potential evoked by faces. Exp Brain Res 96:163–172
Jiang F, Blanz V, O’Toole AJ (2006) Probing the visual representation of faces with adaptation: a view from the other side of the mean. Psychol Sci 17:493–500
Kaspar K, Hassler U, Martens U, Trujillo-Barreto N, Gruber T (2010) Steady-state visually evoked potential correlates of object recognition. Brain Res 1343:112–121
Keil A, Gruber T, Müller MM, Moratti S, Stolarova M, Bradley MM, Lang PJ (2003) Early modulation of visual perception by emotional arousal: evidence from steady-state visual evoked brain potentials. Cogn Affect Behav Neurosci 3:195–206
Keil A, Ihssen N, Heim S (2006) Early cortical facilitation for emotionally arousing targets during the attentional blink. BMC Biol 20(4):23
Kimura M, Schröger E, Czigler I (2011) Visual mismatch negativity and its importance in visual cognitive sciences. NeuroReport 22:669–673
Klimesch W, Doppelmayr M, Russegger H, Pachinger T (1996) Theta band power in the human scalp EEG and the encoding of new information. NeuroReport 7:1235–1240
Klimesch W, Sauseng P, Hanslmayr S (2007) EEG alpha oscillations: the inhibition-timing hypothesis. Brain Res Rev 53:63–88
Kohn A (2007) Visual adaptation: physiology, mechanisms, and functional benefits. J Neurophysiol 97:3155–3164
Kovács G, Zimmer M, Bankó E, Harza I, Antal A, Vidnyánszky Z (2006) Electrophysiological correlates of visual adaptation to faces and body parts in humans. Cereb Cortex 16:742–753
Lalor EC, Pearlmutter BA, Reilly RB, McDarby G, Foxe JJ (2006) The VESPA: a method for the rapid estimation of a visual evoked potential. Neuroimage 32:1549–1561
Lalor EC, Kelly SP, Foxe JJ (2012) Generation of the VESPA response to rapid contrast fluctuations is dominated by striate cortex: evidence from retinotopic mapping. Neuroscience 218:226–234
LeGrand R, Mondloch CJ, Maurer D, Brent HP (2001) Neuroperception. Early visual experience and face processing. Nature 410:890
Leopold DA, Bondar IV, Giese MA (2006) Norm-based face encoding by single neurons in the monkey inferotemporal cortex. Nature 442:572–575
Liebe S, Hoerzer GM, Logothetis NK, Rainer G (2012) Theta coupling between V4 and prefrontal cortex predicts visual short-term memory performance. Nat Neurosci 15:456–462
Liu-Shuang J, Norcia AM, Rossion B (2014) An objective index of individual face discrimination in the right occipito-temporal cortex by means of fast periodic visual stimulation. Neuropsychologia 52:57–72
Luck SJ (2005) An introduction to the event-related potential technique. MIT Press, Cambridge
Makeig S, Jung TP, Bell AJ, Ghahremani D, Sejnowski TJ (1997) Blind separation of auditory event-related brain responses into independent components. Proc Natl Acad Sci USA 94:10979–10984
Malpass RS, Kravitz J (1969) Recognition for faces of own and other race. J Pers Soc Psychol 13:330–334
Maurer D, Grand RL, Mondloch CJ (2002) The many faces of configural processing. Trends Cogn Sci 6:255–260
McTeague LM, Shumen JR, Wieser MJ, Lang PJ, Keil A (2011) Social vision: sustained perceptual enhancement of affective facial cues in social anxiety. Neuroimage 54:1615–1624
Miller EK, Li L, Desimone R (1991) A neural mechanism for working and recognition memory in inferior temporal cortex. Science 254:1377–1379
Mondloch CJ, Geldart S, Maurer D, Le Grand R (2003) Developmental changes in face processing skills. J Exp Child Psychol 86:67–84
Moratti S, Keil A, Stolarova M (2004) Motivated attention in emotional picture processing is reflected by activity modulation in cortical attention networks. Neuroimage 21:954–964
Morgan ST, Hansen JC, Hillyard SA (1996) Selective attention to stimulus location modulates the steady-state visual evoked potential. Proc Natl Acad Sci USA 93:4770–4774
Mouraux A, Iannetti GD (2008) Across-trial averaging of event-related EEG responses and beyond. Magn Reson Imaging 26:1041–1054
Muller MM, Teder W, Hillyard SA (1997) Magnetoencephalographic recording of steady-state visual evoked cortical activity. Brain Topogr 9:163–168
Müller MM, Andersen S, Trujillo NJ, Valdés-Sosa P, Malinowski P, Hillyard SA (2006) Feature-selective attention enhances color signals in early visual areas of the human brain. Proc Natl Acad Sci USA 103:14250–14254
Näätänen R, Gaillard AW, Mäntysalo S (1978) Early selective-attention effect on evoked potential reinterpreted. Acta Psychol 42:313–329
Narici L, Portin K, Salmelin R, Hari R (1998) Responsiveness of human cortical activity to rhythmical stimulation: a three-modality, whole-head neuromagnetic investigation. Neuroimage 7:209–223
Norcia AM, Tyler CW (1985) Spatial frequency sweep VEP: visual acuity during the first year of life. Vision Res 25:1399–1408
Norcia AM, Candy TR, Pettet MW, Vildavski VY, Tyler CW (2002) Temporal dynamics of the human response to symmetry. J Vis 2:132–139
Nunez PL, Srinivasan R (2006) Electrical fields of the brain: the neurophysics of EEG. Oxford University Press, New York
Pazo-Alvarez P, Cadaveira F, Amenedo E (2003) MMN in the visual modality: a review. Biol Psychol 63:199–236
Puce A, Allison T, Bentin S, Gore JC, McCarthy G (1998) Temporal cortex activation in humans viewing eye and mouth movements. J Neurosci 18:2188–2199
Regan D (1966) Some characteristics of average steady-state and transient responses evoked by modulated light. Electroencephalogr Clin Neurophysiol 20:238–248
Regan D (1973) Rapid objective refraction using evoked brain potentials. Invest Ophthalmol 12:669–679
Regan D (1974) Electrophysiological evidence for colour channels in human pattern vision. Nature 250:437–439
Regan D (1977) Steady-state evoked potentials. J Opt Soc Am 67:1475–1489
Regan D (1989) Human brain electrophysiology: evoked potentials and evoked magnetic fields in science and medicine. Elsevier, New York
Ringo JL (1996) Stimulus specific adaptation in inferior temporal and medial temporal cortex of the monkey. Behav Brain Res 76:191–197
Rolls ET, Tovee MJ (1995) Sparseness of the neuronal representation of stimuli in the primate temporal visual cortex. J Neurophysiol 73:713–726
Rossion B (2008) Picture-plane inversion leads to qualitative changes of face perception. Acta Psychol 128:274–289
Rossion B (2009) Distinguishing the cause and consequence of face inversion: the perceptual field hypothesis. Acta Psychol 132:300–312
Rossion B (2013) The composite face illusion: a whole window into our understanding of holistic face perception. Vis Cogn 21:139–253
Rossion B, Boremanse A (2011) Robust sensitivity to facial identity in the right human occipito-temporal cortex as revealed by steady-state visual-evoked potentials. J Vis 16:1–21
Rossion B, Jacques C (2008) Does physical interstimulus variance account for early electro-physiological face sensitive responses in the human brain? Ten lessons on the N170. NeuroImage 39:1959–1979
Rossion B, Jacques C (2011) The N170: understanding the time-course of face perception in the human brain. In: Luck S, Kappenman E (eds) The Oxford handbook of ERP components. University Press, Oxford, pp 115–142
Rossion B, Michel C (2011) An experienced-based holistic account of the other-race face effect. In: Calder A, Rhodes G, Haxby JV, Johnson M (eds) The Oxford handbook of face perception. Oxford University Press, Oxford, pp 215–244
Rossion B, Delvenne J-F, Debatisse D, Goffaux V, Bruyer R, Crommelinck M, Guerit J-M (1999) Spatio-temporal brain localization of the face inversion effect. Biol Psychol 50:173–189
Rossion B, Prieto EA, Boremanse A, Kuefner D, Van Belle G (2012) A steady-state visual evoked potential approach to individual face perception: effect of inversion, contrast-reversal and temporal dynamics. NeuroImage 63:1585–1600
Russell R, Sinha P, Biederman I, Nederhouser M (2006) Is pigmentation important for face recognition? Evidence from contrast negation. Perception 356:749–759
Sadr J, Jarudi I, Sinha P (2003) The role of eyebrows in face recognition. Perception 65:285–293
Santarelli R, Maurizi M, Conti G, Ottaviani F, Paludetti G et al (1995) Generation of human auditory steady-state responses (SSRs) II: addition of responses to individual stimuli. Hear Res 83:9–18
Schultz J, Pilz KS (2009) Natural facial motion enhances cortical responses to faces. Exp Brain Res 194:465–475
Schweinberger SR, Pickering EC, Jentzsch I, Burton AM, Kaufmann JM (2002) Event-related brain potential evidence for a response of inferior temporal cortex to familiar face repetitions. Cogn Brain Res 14:398–409
Sergent J (1984) Configural processing of faces in the left and the right cerebral hemispheres. J Exp Psychol 10:554–572
Sergent J, Signoret JL (1992) Varieties of functional deficits in prosopagnosia. Cereb Cortex 2:375–388
Silberstein RB, Schier MA, Pipingas A, Ciorciari J, Wood SR, Simpson DG (1990) Steady-state visually evoked potential topography associated with a visual vigilance task. Brain Topogr 3:337–347
Srinivasan R, Russell DP, Edelman GM, Tononi G (1999) Increased synchronization of neuromagnetic response during conscious perception. J Neurosci 19:5435–5448
Srinivasan R, Bibi FA, Nunez PL (2006) Steady-state visual evoked potentials: distributed local sources and wave-like dynamics are sensitive to flicker frequency. Brain Topogr 18:167–187
Talsma D, Doty TJ, Strowd R, Woldorff MG (2006) Attentional capacity for processing concurrent stimuli is larger across sensory modalities than within a modality. Psychophysiology 43:541–549
Tanaka JW, Farah MJ (1993) Parts and wholes in face recognition. Q J Exp Psychol 46:225–245
Towler J, Eimer M (2012) Electrophysiological studies of face processing in developmental prosopagnosia: neuropsychological and neurodevelopmental perspectives. Cogn Neuropsychol 29:503–529
Tsuruhara A, Inui K, Kakigi R (2014) Steady-state visual-evoked response to upright and inverted geometrical faces: a magnetoencephalography study. Neurosci Lett 562:19–23
Tyler CW, Kaitz M (1977) Movement adaptation in the visual evoked response. Exp Brain Res 27:203–209
Valentine T, Powell J, Davidoff J, Letson S, Greenwood R (2006) Prevalence and correlates of face recognition impairments after acquired brain injury. Neuropsychol Rehabil 16:272–297
Van der Tweel LH, Lunel HF (1965) Human visual responses to sinusoidally modulated light. Electroencephalogr Clin Neurophysiol 18:587–598
van Vugt MK, Simen P, Nystrom LE, Holmes P, Cohen JD (2012) EEG oscillations reveal neural correlates of evidence accumulation. Front Neurosci 6:106
Walther C, Schweinberger SR, Kaiser D, Kovács G (2013) Neural correlates of priming and adaptation in familiar face perception. Cortex 49:1963–1977
Wieser MJ, McTeague LM, Keil A (2012) Competition effects of threatening faces in social anxiety. Emotion 12:1050–1060
Wilmer JB, Germine L, Chabris CF, Chatterjee G, Williams M, Loken E, Nakayama K, Duchaine B (2010) Human face recognition ability is specific and highly heritable. Proc Natl Acad Sci USA 107:5238–5241
Yin RK (1969) Looking at upside-down faces. J Exp Psychol 81:141–145
Young MP, Yamane S (1992) Sparse population coding of faces in IT cortex. Science 256:1327–1331
Young AW, Hellawell D, Hay DC (1987) Configurational information in face perception. Perception 16:747–759
Zemon V, Ratliff F (1982) Visual evoked potentials: evidence for lateral interactions. Proc Natl Acad Sci USA 79:5723–5726
Acknowledgments
This work was supported by the Belgian National Foundation for Scientific Research (FNRS) and an ERC Grant (facessvep 284025).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (MOV 284 kb)
Supplementary material 2 (MOV 988 kb)
Rights and permissions
About this article
Cite this article
Rossion, B. Understanding individual face discrimination by means of fast periodic visual stimulation. Exp Brain Res 232, 1599–1621 (2014). https://doi.org/10.1007/s00221-014-3934-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-014-3934-9