Abstract
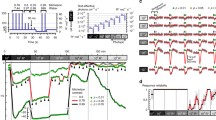
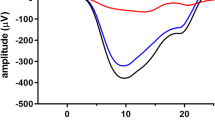
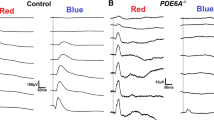
Although gross recordings of the ganzfeld flash-evoked electroretinogram (ERG) can potentially provide information about the activity of many, if not all, retinal cell types, it is necessary to dissect the ERG into its components to realize this potential fully. Here we describe various procedures that have been used in intact mammalian eyes to identify and characterize the contributions to the dark-adapted ERG of different cells in the retinal rod pathway. These include (1) examination of the very early part of the response to a flash (believed to reflect directly the photocurrent of rods), (2) application of high-energy probe flashes to provide information about the underlying rod photoreceptor response even when this component is obscured by the responses of other cells, (3) pharmacological suppression of responses of amacrine and ganglion cells to identify the contribution of these cells and to reveal the weaker responses of bipolar cells, (4) use of pharmacological agents that block transmission of signals from rods to more proximal neurons to separate responses of rods from those of later neurons, (5) examination of the ERG changes produced by ganglion-cell degeneration or pharmacological block of nerve-spike generation to identify the contribution of spiking neurons, (6) modeling measured amplitude-energy functions and timecourse of flash responses and (7) using steady backgrounds to obtain differential reductions in sensitivity of different cell types. While some of these procedures can be applied to humans, the results described here have all been obtained in studies of the ERG of anaesthetized cats, or macaque monkeys whose retinas are very similar to those of humans.
Similar content being viewed by others
References
Smith RG, Freed MA, Sterling P. Microcircuitry of the dark-adapted cat retina: functional architecture of the rod-cone network. J Neurosci 1986; 6: 3505–17.
Sterling PE, Cohen E, Freed MA, Smith RG. Microcircuitry of the on-beta ganglion cell in daylight, twilight and star light. Neurosci Res 1987; 6; s269–s285.
Freed MA, Smith RG, Sterling P. Rod bipolar array in the cat retina: pattern of input from rods and GABA-accumulating amacrine cells. J Comp Neurol 1987; 266: 445–55.
Grünert U, Martin PR. Rod bipolar cells in the macaque monkey retina: immunoreactivity and connectivity. J Neurosci 1991; 11: 2742–58.
Kolb, H, Linberg KA, Fisher SK. Neurons of the human retina: A Golgi study. J Comp Neurol 1992; 318: 147–87.
Wässle H, Grünert U, Chun MH, Boycott BB. The rod pathway of the macaque monkey retina: identification of AII-amacrine cells with antibodies against calretinin. J Comp Neurol 1995; 361: 537–51.
Frishman LJ, Reddy MG, Robson JG. Effects of background light on the human dark-adapted electroretinogram and psychological threshold. J Opt Soc. Soc Am A 1996; 13: 601–12.
Sieving PA, Frishman LJ, Steinberg RH. Scotopic threshold response of proximal retina in cat. J. Neurophysiol 1986; 56: 1049–61.
Steinberg RH, Frishman LJ, Sieving PA. (1991) Negative components of the electroretinogram from proximal retina and photoreceptor. In: NN Osborne and GJ Chader, eds. Progress in Retinal Research. Oxford, UK: Pergamon Press 1990; 10: 121–60.
Newman EA. Current source-density analysis of the b-wave of frog retina. J Neurophysiol 1980; 43: 1355–66.
Newman EA, Odette LL. Model of electroretinogram b-wave generation: a test of the K C hypothesis. J Neurophysiol 1984; 51: 164–82.
Stockton RA, Slaughter MM. b-Wave of the electroretinogram: a reflection of ON bipolar cell activity. J Gen Physiol 1989; 93: 101–22.
Gurevich L, Slaughter MM. Comparison of the waveforms of the ON bipolar neuron and the b-wave of the electroretinogram. Vision Res 1993; 33: 2431–35.
Xu XJ, Karwoski CJ. Current source density analysis of retinal field potentials. 2. Pharmacological analysis of the b-wave and m-wave. J Neurophysiol 1994; 72: 96–105.
Robson JG, Frishman LJ. Response linearity and dynamics of the cat retina: the bipolar cell component of the dark-adapted ERG. Vis. Neurosci 1995; 12: 837–50.
Tian N, Slaughter MM. Correlation of dynamic responses in the ON bipolar neuron and the b-wave of the electroretinogram. Vision Res 1995; 35: 1359–64.
Frishman LJ, Shen FF, Du L, Robson JG, Harwerth RS, Smith III EL, Carter-Dawson U, Crawford MLJ. The scotopic electroretinogram of macaque after retinal ganglion cell loss from experimental glaucoma. Invest Ophthalmol Vis Sci 1996; 37: 125–41.
Breton ME, Montzka DP. Empiric limits of rod photocurrent component underlying a-wave response in the electroretinogram. Doc Ophthalmol 1992; 79: 337–61.
Massey SC. types using glutamate as a neurotransmitter in the vertebrate retina. In: NN Osborne and GJ Chader, eds. Progress in Retinal Research. Oxford, UK: Pergamon Press 1990; 9: 399–425.
Sterling P. Retina. In: G Shepherd ed. Synaptic Organization of the Brain. Oxford, UK: Oxford Univ. Press, chapt. 6, 1990.
Wässle H, Boycott BB. Functional architecture of the mammalian retina. Physiol Rev 1991; 71: 447–80.
Robson JG, Frishman LJ. Photoreceptor and bipolar-cell contributions to the cat elec-troretinogram: a kinetic model for the early part of the flash response. J Opt Soc Am A 1996; 13: 613–22.
Dawson WW, Trick GL, Litzkow CA. Improved electrode for electroretinography. Invest. Ophthalmol Vis Sci 1979; 18: 988–91.
Lamb TD, McNaughton PA, Yau KW. Spatial spread of activation and background desensitization in toad rod outer segments. J Physiol (Lond) 1981; 319: 463–96.
Baylor DA, Nunn BJ, Schnapf JL. The photocurrent, noise and spectral sensitivity of rods of the monkey macaca fascicularis. J Physiol (Lond) 1984; 357: 575–607.
Tamura T, Nakatani K, Yau K-W. Light adaptation in cat retinal rods. Science 1989; 245: 755–58.
Hood DC, Birch DG. Light adaptation of human rod receptors: the leading edge of the human a-wave and models of rod receptor activity. Vision Res 1993; 33: 1605–18.
Penn RD, Hagins WA. Kinetics of the photocurrent of retinal rods. Biophys J 1972; 12: 1073–94.
Breton ME, Schueller AW, Lamb TD, Pugh EN, Jr. Analysis of ERG a-wave amplification and kinetics in terms of the G-protein cascade of phototransduction. Invest Ophthalmol & Vis Sci 1994; 35: 295–309.
Lamb TD, Pugh EN. A quantitative account of the activation steps involved in phototransduction in amphibian photoreceptors. J. Physiol 1992; 449: 717–57.
Smith NP, Lamb TD. The a-wave of the human electroretinogram recorded with a minimally invasive technique. Vision Res 1997; 37: 2943–52.
Kraft TW, Schneeweis DM, Schnapf JL. Visual transduction in human rod photoreceptors. J Physiol (Lond) 1993; 464: 747–65.
Pepperberg DR, Birch DG, Hood DC. Photoresponses of human rods in vivo. Visual Neurosci 1997; 14: 73–82..215
Frishman LJ, Sieving PA. Evidence for 2 sites of adaptation affecting the dark-adapted ERG of cats and primates. Vision Res 1995; 35: 435–42.
Hood DC, Birch DG. (1996) The b-wave of the scotopic (rod) ERG as a measure of the activity of human on-bipolar cells. J Opt Soc Am A 1996; 13: 623–33.
Vaegan, Millar TJ. Effect of kainic acid and NMDA on the pattern electroretinogram, the scotopic threshold response, the oscillatory potentials and the electroretinogram in the urethane anaesthetized cat. Vision Res 1994; 34: 1111–25.
Barlow HB, Levick WR, Yoon M. Responses to single quanta of light in retinal ganglion cells of the cat. Vision Res Suppl 1971; 3: 87–101.
Mastronarde DN. Correlated firing of cat retinal ganglion cells. II. Responses of X-and Y-cells to single quantal events. J Neurophysiol 1983; 49: 325–49.
Hood DC, Birch DG. A computational model of the amplitude and implicit time of the b-wave of the human ERG. Vis Neurosci 1992; 8: 107–26.
Shiells RA, Falk G. Glutamate receptors of rod bipolar cells are linked to a cyclic GMP cascade via a G-protein. Proc Roy Soc Lond, Series B: Bio Sci 1990; 242: 91–4.
Nawy 5, Jahr CE. Suppression by glutamate of cGMP-activated conductance in retinal bipolar cells. Nature 1990; 346: 269–71.
Ashmore JF, Falk G. The single-photon signal in rod bipolar cells of the dogfish retina. J Physiol (Lond) 1980; 300: 151–66.
Miller RF, Dowling JE. Intracellular responses of the Müller (glial) cells of mudpuppy retina: their relation to b-wave of the electroretinogram. J Neurophysiol 1970; 33: 323–41.
Wen R, Oakley B. K+-evoked Müller cell depolarization generates b-wave of electroret-inogram in toad retina. Proc Nat Acad Sci (USA) 1990; 87: 2117–21.
Wakabayashi K, Gieser J, Sieving PA. Aspartate separation of the scotopic threshold response (STR) from the photoreceptor a-wave of the cat and monkey ERG. Invest Ophthalmol Vis Sci 1988; 29: 1615–22.
Frishman LJ, Steinberg RH. Intraretinal analysis of the threshold dark-adapted ERG of cat retina. J Neurophysiol 1989; 61: 1221–32.
Frishman LJ, Steinberg RH. Light-evoked increases in [K C ]o in proximal portion of the dark-adapted cat retina. J Neurophyisol 1989; 61: 1233–43.
Sieving PA. Retinal ganglion cell loss does not abolish the STR of the cat and human ERG. Clin Vision Sci 1991; 6: 149–58.
Frishman LJ, Robson JG, Du L. Contributions of the positive and negative scotopic threshold responses to the scotopic cat ERG. Invest Ophthalmol Vis Sci Suppl 1993; 34: 1273.
Robson JG, Frishman LJ, Du L. Component potentials of the dark-adapted cat ERG. Soc Neurosci Abs 1993; 1413.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Robson, J.G., Frishman, L.J. Dissecting the dark-adapted electroretinogram. Doc Ophthalmol 95, 187–215 (1998). https://doi.org/10.1023/A:1001891904176
Issue Date:
DOI: https://doi.org/10.1023/A:1001891904176