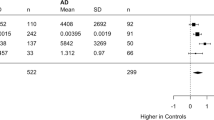
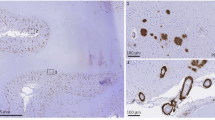
Abstract
Fibrillar amyloid β-protein has been implicated in the pathogenesis of Alzheimer's disease because of its neurotoxicity and its ability to activate complement1–3. Reactive microglia, astrocytes4,5 and complement (C') components (reviewed in ref. 6) are associated with senile plaques, the fibrillar, β-sheet assemblies of amyloid β-peptide found predominantly in brain from individuals with AD (ref. 7). These indications of inflammatory events are not prevalent in the nonfibrillar “diffuse” plaques often seen in age-matched control cases without dementia. Clinical studies over the past several years have correlated the use of anti-inflammatory drugs with a decrease in the incidence and progression of AD dementia and/or dysfunction, supporting a role for gliosis and inflammation in AD pathogenesis8,9 (reviewed in ref. 6). C5a, a product of C' activation, is chemotactic for microglia10. Thus, complement activation provides a specific mechanism for recruiting reactive glial cells to the site of the fibrillar amyloid β-protein plaque, which could lead to inflammatory events, neuronal dysfunction and degeneration5. With the use of truncated amyloid β-peptides, the region of amyloid β-protein limited by residues 4 and 11 has been identified as critical in the interaction between amyloid β-protein and C1q, the recognition component of the classical complement pathway (CCP), which results in the activation of C'. Furthermore, substitution of an isoaspartic acid for aspartic acid at amyloid β-protein residue 7 resulted in the complete elimination of CCP-activating activity. A molecular model of this interaction has been generated that should be useful in the design of candidate therapeutic inhibitors of CCP activation by amyloid β-protein.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Hsiao, K.K. et al. Correlative memory deficits, Ap elevations, and amyloid plaques in transgenic mice. Science 274, 99–102 (1996).
Selkoe, D.J. The molecular pathology of Alzheimer's disease. Neuron 6, 487–498 (1991).
Jiang, H., Burdick, D., Glabe, C.G., Cotman, C.W. & Tenner, A.J. β-Amyloid activates complement by binding to a specific region of the collagen-like domain of the C1q A chain. J.Immunol. 152, 5050–5059 (1994).
Maat-Schieman, M.L.C. et al. Microglia in diffuse plaques in hereditary cerebral hemorrhage with amyloidosis (Dutch): An immunohistochemical study. J. Neuropathol. Exp. Neurol. 5, 483–491 (1994).
Cotman, C.W., Tenner, A.J. & Cummings, B.J. β-Amyloid converts an acute phase injury response to chronic injury responses. Neurobiol. Aging 17, 723–731 (1996).
Pasinetti, G.M. Inflammatory mechanisms in neurodegeneration and Alzheimer's disease: The role of the complement system. Neurobiol. Aging 17, 707–716 (1996).
Afagh, A., Cummings, B.J., Cribbs, D.H., Cotman, C.W. & Tenner, A.J. Localization and cell association of C1q in Alzheimer's disease brain. Exp. Neurol. 138, 22–32 (1996).
Breitner, J.C.S. et al. Delayed onset of Alzheimer's disease with nonsteroidal anti-inflammatory and histamine H2 blocking drugs. Neurobiol. Aging 4, 523–530 (1995).
McGeer, P.L., McGeer, E., Rogers, J. & Sibley, J. Anti-inflammatory drugs and Alzheimer disease. Lancet 335, 1037 (1990).
Yao, J., Harvath, L., Gilbert, D.L. & Colton, C.A. Chemotaxis by a CNS macrophage, the microglia. J. Neurosci. Res. 27, 36–42 (1990).
Pike, C.J., Overman, M.J. & Cotman, C.W. Amino-terminal deletions enhance aggregation of β-amyloid peptides in vitro. J. Biol. Chem. 270, 23895–23898 (1995).
Hilbich, C., Kisters-Woike, B., Reed, J., Masters, C.L. & Beyreuther, K. Human and rodent sequence analogs of Alzheimer's amyloid beta A4 share similar properties and can be solubilized in buffers of pH 7.4. Eur. J. Biochem. 201, 61–69 (1991).
Johnson, B.A. et al. Protein L-isoaspartyl methyltransferse in postmortem brains of aged humans. Neurobiol. Aging 12, 19–24 (1991).
Roher, A.E. et al. Structural alterations in the peptide backbone of beta-amyloid core protein may account for its deposition and stability in Alzheimer's disease. J. Biol. Chem. 268, 3072–3083 (1993).
Bella, J., Eaton, M., Brodsky, B. & Berman, H.M. Crystal and molecular structure of a collagen-like peptide at 1.9 angstrom resolution. Science 266, 75–81 (1994).
Nomizu, M. et al. The all-D-configuration segment containing the IKVAV sequence of laminin A chain has similar activities to the all-L-peptide in vitro and in vivo. J. Biol. Chem. 267, 14118–14121 (1992).
Rogers, J. et al. Complement activation by beta-amyloid in Alzheimer disease. Proc. Natl. Acad. Sci. USA 89, 10016–10020 (1992).
Yao, J., Keri, J.E., Taffs, R.E. & Colton, C.A. Characterization of interleukin-1 production by microglia in culture. Brain Res. 591, 88–93 (1992).
Gasque, P., Fontaine, M. & Morgan, B.P. Complement expression in human brain: Biosynthesis of terminal pathway components and regulators in human glial cells and cell lines. J. Immunol. 154, 4726–4733 (1995).
Colton, C.A. & Gilbert, D.L. Microglia, an in vivo source of reactive oxygen species in the brain. Adv. Neurol. 59, 321–326 (1993).
Meda, L. et al. Activation of microglial cells by β-amyloid protein and interferon-gamma. Nature 374, 647–650 (1995).
El Khoury, J. et al. Scavenger receptor-mediated adhesion of microglia to β-amyloid fibrils. Nature 382, 716–719 (1996).
Paresce, D., Ghosh, R.N. & Maxfield, F.R. Microglial cells internalize aggregates of the Alzheimer's disease amyloid β-protein via a scavenger receptor. Neuron 17, 553–565 (1996).
Yan, S.D. et al. RAGE and amyloid-β peptide neurotoxicity in Alzheimer's disease. Nature 382, 685–691 (1996).
Kilchherr, E., Hofmann, H., Steigemann, W. & Engel, J. Structural model of the collagen-like region of C1q comprising the kink region and the fibre-like packing of the six triple helices. J. Mol. Biol. 186, 403–416 (1985).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Velazquez, P., Cribbs, D., Poulos, T. et al. Aspartate residue 7 in amyloid β-protein is critical for classical complement pathway activation: Implications for Alzheimer's disease pathogenesis. Nat Med 3, 077–079 (1997). https://doi.org/10.1038/nm0197-77
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0197-77
This article is cited by
-
As a Potential Therapeutic Target, C1q Induces Synapse Loss Via Inflammasome-activating Apoptotic and Mitochondria Impairment Mechanisms in Alzheimer’s Disease
Journal of Neuroimmune Pharmacology (2023)
-
The good, the bad, and the opportunities of the complement system in neurodegenerative disease
Journal of Neuroinflammation (2020)
-
Suche nach Risikogenen bei der Alzheimer-Erkrankung
Der Nervenarzt (2017)
-
Age-related increases in amyloid beta and membrane attack complex: evidence of inflammasome activation in the rodent eye
Journal of Neuroinflammation (2015)
-
Genetics of Alzheimer's Disease
Neurotherapeutics (2014)