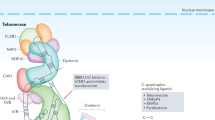
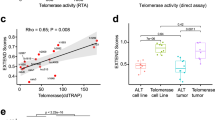
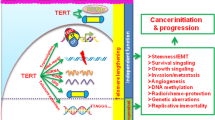
Abstract
The role of telomerase in actively proliferating cells is assumed to be restricted to maintaining of telomeres above a minimum-length threshold, thereby preventing telomere exhaustion and chromosomal instability. However, forced telomerase expression in cells and mice with normal-length telomeres has shown that telomerase promotes growth and survival in a manner that is uncoupled from net telomere lengthening. These findings imply that telomerase might have a fundamental role in tumour growth and survival, even at stages when telomeres are sufficiently long.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Blackburn, E. H. Switching and signaling at the telomere. Cell 106, 661–673 (2001).
Chan, S.W-L. & Blackburn, E. H. New ways not to make ends meet: telomerase, DNA damage proteins and heterochromatin. Oncogene 21, 553–563 (2002).
De Lange, T. . Protection of mammalian telomeres. Oncogene 21, 532–540 (2002).
Griffith, J. D. et al. Mammalian telomeres end in a large duplex loop. Cell 97, 503–514 (1999).
Ancelin, K. et al. Targeting assay to study the cis functions of human telomeric proteins: evidence for inhibition of telomerase by TRF1 and for activation of telomere degradation by TRF2. Mol. Cell. Biol. 22, 3474–3487 (2002).
Karlseder, J., Smogorzewska, A. and de Lange, T. Senescence induced by altered telomere state, not telomere loss. Science 295, 2446–2449 (2002).
Goytisolo, F. A. & Blasco, M. A. Many ways to telomere dysfunction: in vivo studies using mouse models. Oncogene 21, 584–591 (2002).
Blasco, M. A. et al. Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell 91, 25–34 (1997).
Espejel, S. et al. Mammalian Ku86 mediates chromosomal fusions and apoptosis caused by critically short telomeres. EMBO J. 21, 2207–2219 (2002).
Harley, C. B., Futcher, A. B. & Greider, C. W. Telomeres shorten during ageing of human fibroblasts. Nature 345, 458–460 (1990).
Collins, K. & Mitchell, J. R. Telomerase in the human organism. Oncogene 21, 564–579 (2002).
Lee, H-W. et al. Essential role of mouse telomerase in highly proliferative organs. Nature 392, 569–574 (1998).
Samper, E., Flores, J. M. & Blasco, M. A. Restoration of telomerase activity rescues chromosomal instability and premature aging in Terc−/− mice with short telomeres. EMBO Rep. 2, 800–807 (2001).
Hemann, M. T., Strong, M. A., Hao, L. Y. and Greider, C. W. The shortest telomere, not average telomere length, is critical for cell viability and chromosome stability. Cell 107, 67–77 (2001).
Bodnar, A. G. et al. Extension of life-span by introduction of telomerase into normal human cells. Science 279, 349–352 (1998).
Hiyama, E. & Hiyama, K. Clinical utility of telomerase in cancer. Oncogene 21, 643–649 (2002).
Mattson, M. P., Fu, W. & Zhang, P. Emerging roles for telomerase in regulating cell differentiation and survival: a neuroscientist's perspective. Mech. Aging Dev. 122, 659–671 (2001).
González-Suárez, E. et al. Increased epidermal tumors and increased wound healing in transgenic mice overexpressing the catalytic subunit of telomerase, mTERT, in basal keratinocytes. EMBO J. 20, 2619–2630 (2001).
Reyes, M. et al. Purification and ex vivo expansion of postnatal human marrow mesodermal progenitor cells. Blood 98, 2615–2625 (2001).
McEachern, M. J. & Blackburn, E. H. Runaway telomere elongation caused by telomerase RNA gene mutations. Nature 376, 403–409 (1995).
Smogorzewska, A. et al. Control of human telomere length by TRF1 and TRF2. Mol. Cell. Biol. 20, 1659–1668 (2000).
Prescott, J. C. & Blackburn, E. H. Telomerase RNA template mutations reveal sequence-specific requirements for activation and repression of telomerase action at telomeres. Mol. Cell. Biol. 20, 2941–2948 (2000).
Herrera, E. et al. Disease states associated to telomerase deficiency appear earlier in mice with short telomeres. EMBO J. 18, 2950–2960 (1999).
Hastie, N. D. et al. Telomere reduction in human colorectal carcinoma and with ageing. Nature 346, 866–868 (1990).
de Lange, T. et al. Structure and variability of human chromosome ends. Mol. Cell. Biol. 10, 518–527 (1990).
Zhu, J., Wang, H., Bishop, M. and Blackburn, E. H. Telomerase extends the lifespan of virus-transformed human cells without net telomere lengthening. Proc. Natl Acad. Sci. USA 96, 3723–3728 (1999).
Chin, L. et al. p53 deficiency rescues the adverse effects of telomere loss and cooperates with telomere dysfunction to accelerate carcinogenesis. Cell 97, 527–538 (1999).
González-Suárez, E., Samper, E., Flores, J. M. & Blasco, M. A. Telomerase-deficient mice with short telomeres are resistant to skin tumorigenesis. Nature Genet. 26, 114–117 (2000).
Corey, D. R. Telomerase inhibition, oligonucleotides, and clinical trials. Oncogene 21, 631–727 (2002).
Neidle, S. & Read, M. A. G-quadruplexes as therapeutic targets. Biopolymers 56, 195–208 (2000).
Damm, K. et al. A highly selective telomerase inhibitor limiting human cancer cell proliferation. EMBO J. 20, 6958–6968 (2001).
Blasco, M. A., Rizen, M., Greider, C. W. and Hanahan, D. Differential regulation of telomerase activity and its RNA component during multistage tumorigenesis. Nature Genet. 12, 200–204 (1996).
Broccoli, D., Godley, L. A., Donehower, L. A., Varmus, H. E. & de Lange, T. Telomerase activation in mouse mammary tumors: lack of detectable telomere shortening and evidence for regulation of telomerase RNA with cell proliferation. Mol. Cell. Biol. 16, 3765–3772 (1996).
González–Suárez, E., Flores, J. M. & Blasco, M. A. Cooperation between p53 mutation and high telomerase transgenic expression in spontaneous cancer development. Mol. Cell. Biol. (in the press).
Artandi, S. E. et al. Constitutive telomerase expression promotes mammary carcinomas in aging mice. Proc. Natl Acad. Sci. USA 99, 8191–8196 (2002).
Thomas, M. et al. Cooperation between hTERT, SV40 T antigen and oncogenic Ras in tumorigenesis: a cell transplantation model using bovine adrenocortical cells. Neoplasia (in the press).
Oh, H. et al. Telomerase reverse transcriptase promotes cardiac muscle cell proliferation, hypertrophy, and survival. Proc. Natl Acad. Sci. USA 98, 10308–11033 (2001).
Stampfer, M. R. et al. Expression of the telomerase catalytic subunit, hTERT, induces resistance to transforming growth factor-β growth inhibition in p16INK4A(−) human mammary epithelial cells. Proc. Natl Acad. Sci. USA 98, 4498–4503 (2001).
Lu, C. L., Fu, W. & Mattson, M. P. Telomerase protects developing neurons against DNA damage-induced cell death. Brain Res Dev Brain Res 131, 167–171 (2001).
Zhu, H., Fu, W. & Mattson, M. P. The catalytic subunit of telomerase protects neurones against amyloid β-peptide induced apoptosis. J. Neurochem. 75, 117–124 (2000).
Xiang, H. et al. Human telomerase accelerates growth of lens epithelial cells through regulation of the genes mediating Rb/E2F pathway. Oncogene 21, 3784–3791 (2002).
Liu, Y. et al. The telomerase reverse transcriptase is limiting and necessary for telomerase function in vivo. Curr. Biol. 10, 1459–1462 (2000).
Nikaido, R. et al. Presence of telomeric G-strand tails in the telomerase catalytic subunit Tert knockout mice. Genes Cells 4, 563–572 (1999).
Bailey, S. M., Conforth, M. N., Kurimasa, A., Chen, D. J. and Goodwin, E. H. Strand-specific postreplicative processing of mammalian telomeres. Science 293, 2462–2465 (2001).
Goytisolo, F. A. et al. Short telomeres result in organismal hypersensitivity to ionizing radiation in mammals. J. Exp. Med. 192, 1625–1636 (2000).
Acknowledgements
We thank M. Serrano for critical reading of the manuscript. Research at the laboratory of M.A.B. is funded by the MCYT (Ministry of Science and Technology), CAM (Regional Government of Madrid), the European Union and the DIO (Department of Immunology and Oncology). The DIO was founded and is supported by the Spanish Research Council (CSIC) and by Pharmacia.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Blasco, M. Telomerase beyond telomeres. Nat Rev Cancer 2, 627–633 (2002). https://doi.org/10.1038/nrc862
Issue Date:
DOI: https://doi.org/10.1038/nrc862
This article is cited by
-
The telomere complex and the origin of the cancer stem cell
Biomarker Research (2021)
-
Extraordinary diversity of telomeres, telomerase RNAs and their template regions in Saccharomycetaceae
Scientific Reports (2021)
-
Mechanism and significance of apoptosis of the immortalized human oral mucosal epithelial cells established by Lentivirus-mediated hTERT
Molecular Biology Reports (2020)
-
Telomerase promotes efficient cell cycle kinetics and confers growth advantage to telomerase-negative transformed human cells
Oncogene (2012)
-
Inhibition of telomerase activity preferentially targets aldehyde dehydrogenase-positive cancer stem-like cells in lung cancer
Molecular Cancer (2011)