Abstract
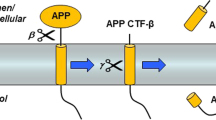
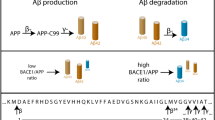
Data that have accumulated for well over a decade have implicated the β-amyloid (Aβ) peptide as a central player in the pathogenesis of Alzheimer’s disease (AD). Amyloid plaques, composed primarily of Aβ progressively form in the brains of AD patients, and mutations in three genes (amyloid precursor protein [APP] and presenilin 1 and 2 [PS1 and PS2]) cause early-onset familial AD (FAD) by directly increasing production of the toxic, plaque-promoting Aβ42 peptide. Given the strong association between Aβ and AD, it is likely that therapeutic strategies to lower the levels of Aβ in the brain should prove beneficial for the treatment of AD. One such strategy could involve inhibiting the enzymes that generate Aβ. Aβ is a product of catabolism of the large type-I membrane protein APP. Two proteases, called β- and γ-secretase, endoproteolyze APP to liberate the Aβ peptide. Recently, the molecules responsible for these proteolytic activities have been identified. Several lines of evidence suggest that the PS1 and PS2 proteins are γ-secretase, and the identity of β-secretase has been shown to be the novel transmembrane aspartic protease, β-site APP-cleaving enzyme 1 (BACE1; also called Asp2 and memapsin 2). BACE2, a protease homologous to BACE1, was also identified, and together the two enzymes define a new family of transmembrane aspartic proteases. BACE1 exhibits all the functional properties of β-secretase, and as the key enzyme that initiates the formation of Aβ, BACE1 is an attractive drug target for AD. This review discusses the identification and initial characterization of BACE1 and BACE2, and summarizes recent studies of BACE1 knockout mice that have validated BACE1 as the authentic β-secretase in vivo.
Similar content being viewed by others
References
Acquati F., Accarino M., Nucci C., Fumagalli P., Jovine L., Ottolenghi S., and Taramelli R. (2000) The gene encoding DRAP (BACE2), a glycosylated transmembrane protein of the aspartic protease family, maps to the down critical region. FEBS Lett. 468, 59–64.
Bennett B. D., Babu-Khan S., Loeloff R., Louis J.-C., Curran E., Citron M., and Vassar R. (2000a) Expression analysis of BACE2 in brain and peripheral tissues. J. Biol. Chem. 275, 20647–20651.
Bennett B. D., Denis P., Haniu M., Teplow D. B., Kahn S., Louis J.-C., et al. (2000b) A furin-like convertase mediates propeptide cleavage of BACE, the Alzheimer’s β-secretase. J. Biol. Chem. 275, 37712–37717.
Bodendorf U., Fischer F., Bodian D., Multhaup G., and Paganetti P. (2001) A splice variant of β-secretase deficient in the amyloidogenic processing of the amyloid precursor protein. J. Biol. Chem. 276, 12019–12023.
Buxbaum J. D., Liu K. N., Luo Y., Slack J. L., Stocking K. L., Peschon J. J., et al. (1998) Evidence that tumor necrosis factor alpha converting enzyme is involved in regulated alpha-secretase cleavage of the Alzheimer amyloid protein precursor. J. Biol. Chem. 273, 27765–27767.
Cai H., Wang Y., McCarthy D., Wen H., Borchelt D. R., Price D. L., and Wong P. C. (2001) BACE1 is the major β-secretase for generation of Aβ peptides by neurons. Nat. Neurosci. 4, 233–234.
Citron M., Diehl T. S., Capell A., Haass C., Teplow D. B., and Selkoe D. J. (1996) Inhibition of amyloid β-protein production in neural cells by the serine protease inhibitor AEBSF. Neuron 17, 171–179.
Citron M., Teplow D. B., and Selkoe D. J. (1995) Generation of amyloid β-protein from its precursor is sequence specific. Neuron 14, 661–670.
Farzan M., Schnitzler C. E., Vasilieva N., Leung D., and Choe H. (2000) BACE2, a β-secretase homolog, cleaves at the β site and within the amyloid-β region of the amyloid-β precursor protein. Proc. Natl. Acad. Sci. USA 97, 9712–9717.
Gandy S. and Greengard P. (1992) Amyloidogenesis in Alzheimer’s disease: Some possible therapeutic opportunities. Trends Pharmacol. Sci. 13, 108–113.
Glenner G. G. and Wong C. W. (1984) Alzheimer’s disease: initial report of the purification and characterization of a novel cerebrovascular amyloid protein. Biochem. Biophys. Res. Commun. 120, 885–890.
Gouras G. K., Xu H., Jovanovic J. N., Buxbaum J. D., Wang R., Greengard P., et al. (1998) Generation and regulation of β-amyloid peptide variants by neurons. J. Neurochem. 71, 1920–1925.
Haass C., Capell A., Citron M., Teplow D. B., and Selkoe D. J. (1995a) The vacuolar H+ ATPase inhibitor bafilomycin A1 differentially affects proteolytic processing of mutant and wild-type β-amyloid precursor protein. J. Biol. Chem. 270, 6186–6192.
Haass C., Hung A. Y., Schlossmacher M. G., Teplow D. B., and Selkoe D. J. (1993) β-Amyloid peptide and a 3-kDa fragment are derived by distinct cellular mechanisms. J. Biol. Chem. 268, 3021–3024.
Haass C., Lemere C. A., Capell A., Citron M., Seubert P., Schenk D., et al. (1995b) The Swedish mutation causes early-onset Alzheimer’s disease by β-secretase cleavage within the secretory pathway. Nat. Med. 1, 1291–1296.
Haass C., Schlossmacher M. G., Hung A. Y., Vigo-Pelfrey C., Mellon A., Ostaszewski B. L., et al. (1992) Amyloid β-peptide is produced by cultured cells during normal metabolism. Nature 359, 322–325.
Haniu M., Denis P., Young Y., Mendiaz E. A., Fuller J., Hui J. O., et al. (2000) Characterization of Alzheimer’s β-secretase protein BACE. J. Biol. Chem. 275, 21099–21106.
Hsiao K., Chapman P., Nilsen S., Eckman C., Harigaya Y., Younkin S., et al. (1996) Correlative memory deficits, Aβ elevation, and amyloid plaques in transgenic mice. Science 274, 99–103.
Hussain I., Powell D. J., Howlett D. R., Chapman G. A., Gilmour L., Murdock P. R., et al. (2000) ASP1 (BACE2) cleaves the amyloid precursor protein at the β-secretase site. Mol. Cell. Neurosci. 16, 609–619.
Hussain I., Powell D., Howlett D. R., Tew D. G., Meek T. D., Chapman C., et al. (1999) Identification of a novel aspartic protease (Asp 2) as β-Secretase. Mol. Cell. Neurosci. 14, 419–427.
Hutton M., Perez-Tur J., and Hardy J. (1998) Genetics of Alzheimer’s disease. Essays Biochem. 33, 117–131.
Kang J., Lemaire H.- G., Unterbeck A., Salbaum J. M., Masters C. L., Grzeschik K.- H., et al. (1987) The precursor of Alzheimer’s disease amyloid A4 protein resembles a cell-surface receptor. Nature 325, 733–736.
Knops J., Suomensaari S., Lee M., McConlogue L., Seubert P., and Sinha S. (1995) Cell-type and amyloid precursor protein-type specific inhibition of Aβ release by bafilomycin A1, a selective inhibitor of vacuolar ATPases. J. Biol. Chem. 270, 2419–2422.
Koo E. H. and Squazzo S. (1994) Evidence that production and release of amyloid β-protein involves the endocytic pathway. J. Biol. Chem. 269, 17386–17389.
Lammich S., Kojro E., Postina R., Gilbert S., Pfeiffer R., Jasionowski M., et al. (1999) Constitutive and regulated alpha-secretase cleavage of Alzheimer’s amyloid precursor protein by a disintegrin metalloprotease. Proc. Natl. Acad. Sci. USA 96, 3922–3927.
Lee V. M.- Y., Balin B. J., Otvos L., and Trojanowski J. Q. (1991) A68. A major subunit of paired helical filaments and derivatized forms of normal tau. Science 251, 675–678.
Lin X., Koelsch G., Wu S., Downs D., Dashti A., and Tang J. (2000) Human aspartic protease memapsin 2 cleaves the β-secretase site of β-amyloid precursor protein. Proc. Natl. Acad. Sci. USA 97, 1456–1460.
Luo, Y. Bolon, B. Kahn, S. Bennett, B. D. Babu-Khan, S. Denis, P., et al. (2001) Mice deficient in BACE1, the Alzheimer’s β-secretase, have normal phenotype and abolished β-amyloid generation. Nat. Neurosci. 4, 231–232.
Marcinkiewicz M., and Seidah N. G. (2000) Coordinated expression of β-amyloid precursor protein and the putative β-secretase BACE and α-secretase ADAM10 in mouse and human brain. J. Neurochem. 75, 2133–2143.
Mullan M., Crawford F., Houlden H., Axelman K., Lilius L., Winblad B., and Lannfelt L. (1992a) A pathogenic mutation for probable Alzheimer’s disease in the APP gene at the N-terminus of β-amyloid. Nat. Genet. 1, 345–347.
Murphy T., Yip A., Brayne C., Easton D., Evans J. G., Xuereb J., et al. (2001) The BACE gene: genomic structure and candidate gene study in late-onset Alzheimer’s disease. Neuroreport 12, 631–634.
Pike C. J., Overman M. J., and Cotman C. W. (1995) Aminoterminal deletions enhance aggregation of β-amyloid peptides in vitro. J. Biol. Chem. 270, 23895–23898.
Roberds S. L., Anderson J., Basi G., Bienkowski M. J., Branstetter D. G., Chen K. S., et al. (2001) BACE knockout mice are healthy despite lacking the primary β-secretase activity in brain: implications for Alzheimer’s disease therapeutics. Hum. Mol. Genet. 10, 1317–1324.
Roher A. E., Lowenson J. D., Clarke S., Wolkow C., Wang R., Cotter R. J., et al. (1993) Structural alterations in the peptide backbone of β-amyloid core protein may account for its deposition and stability in Alzheimer’s disease. J. Biol. Chem. 268, 3072–3083.
Saunders A. J., Kim T.- W., Tanzi R. E., Fan W., Bennett B. D., Babu-Khan S., et al. (1999) BACE maps to chromosome 11 and a BACE homolog, BACE2, reside in the obligate Down syndrome region of chromosome 21. Science 286, 1255a.
Selkoe D. J. (2001) Alzheimer’s disease: genes, proteins, and therapy. Physiol. Rev. 81, 741–766.
Seubert P., Oltersdorf T., Lee M. G., Barbour R., Blomqist C., Davis D. L., et al. (1993) Secretion of β-amyloid precursor protein cleaved at the amino-terminus of the β-amyloid peptide. Nature 361, 260–263.
Sinha S., Anderson J. P., Barbour R., Basi G. S., Caccavello R., Davis D., et al. (1999) Purification and cloning of amyloid precursor protein β-secretase from human brain. Nature 402, 537–540.
Sisodia S. S., Kim S. H., and Thinakaran G. (1999) Function and dysfunction of the presenilins. Am. J. Hum. Genet. 65, 7–12.
Solans A., Estivill X., and de La Luna S. (2000) A new aspartyl protease on 21q22.3, BACE2, is highly similar to Alzheimer’s amyloid precursor protein β-secretase. Cytogenet. Cell Genet. 89, 177–184.
Tanzi R. E., Gusella J. F., Watkins P. C., Bruns G. A. B., St. George-Hyslop P. H., Van Keuren M. L., et al. (1987) Amyloid β-protein gene: cDNA, mRNA distribution, and genetic linkage near the Alzheimer locus. Science 235, 880–884.
Terry R. D., Masliah E., and Hansen L. A. (1999) The neuropathology of Alzheimer disease and the structural basis of its cognitive alterations, in Alzheimer Disease, Terry, R. D., Katzman, R., Bick, K. L., and Sisodia, S. S., eds., Lippincott Williams and Wilkins, Philadelphia, PA, pp. 187–206.
Vassar R., Bennett B. D., Babu-Khan S., Kahn S., Mendiaz E. A., Denis P., et al. (1999) β-Secretase cleavage of Alzheimer’s amyloid precursor protein by the transmembrane aspartic protease BACE. Science 286, 735–741.
Vassar R. and Citron M. (2000) Aβ-generating enzymes: recent advances in β- and γ-secretase research. Neuron 27, 419–422.
Wolfe M. S., Angeles J. D. L., Miller D. D., Xia W., and Selkoe D. J. (1999a) Are presenilins intramembrane-cleaving proteases? Implications for the molecular mechanism of Alzheimer’s disease. Biochemistry 38, 11223–11230.
Wolfe M. S., Xia W., Ostaszewski B. L., Diehl T. S., Kimberly W. T., and Selkoe D. J. (1999b) Two transmembrane aspartates in presenilin-1 required for presenilin endoproteolysis and gamma-secretase activity. Nature 398, 513–517.
Yan R., Bienkowski M. J., Shuck M. E., Miao H., Tory M. C., Pauley A. M., et al. (1999) Membrane-anchored aspartyl protease with Alzheimer’s disease β-secretase activity. Nature 402, 533–537.
Yan R., Munzner J. B., Shuck M. E., and Bienkowski M. J. (2001) BACE2 functions as an alternative αsecretase in cells. J. Biol. Chem. 276, 34019–34027.
Younkin S. G. (1998) The role of a beta 42 in Alzheimer’s disease. J. Physiol. (Paris) 92, 289–292.
Zhao J., Paganini L., Mucke L., Gordon M., Refolo L., Carman M., et al. (1996) β-Secretase processing of the β-amyloid precursor protein in transgenic mice is efficient in neurons but inefficient in astrocytes. J. Biol. Chem. 271, 31407–31411.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vassar, R. Bace 1. J Mol Neurosci 23, 105–113 (2004). https://doi.org/10.1385/JMN:23:1-2:105
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/JMN:23:1-2:105